Competencies
- AN25.2: Describe development of pleura, lung and heart (lung development is included in this chapter).
- AN25.4: Describe embryological basis of atrial septal defect, ventricular septal defect, Fallot’s tetralogy, tracheoesophageal fistula (Tracheoesophageal fistula is included in this chapter).
INTRODUCTION
The respiratory system develops from two principal components:
- An epithelial lung bud that arises from the foregut endoderm, and
- Surrounding connective tissues derived from the splanchnic mesoderm.
Formation of the Lung Bud
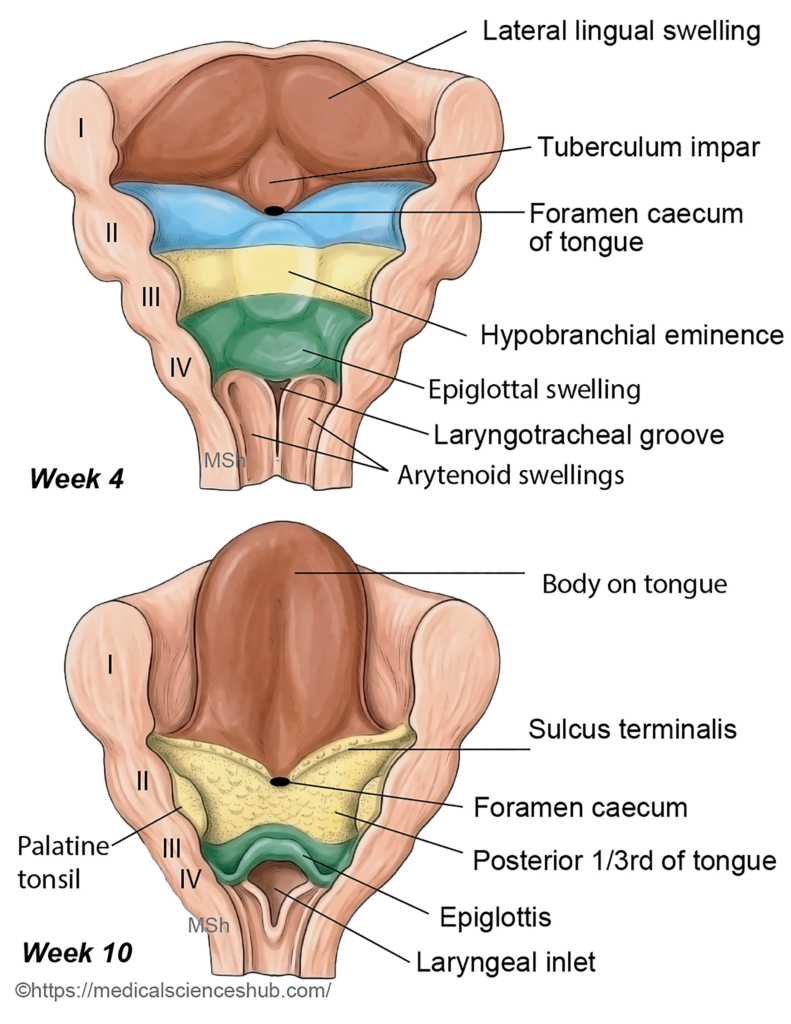
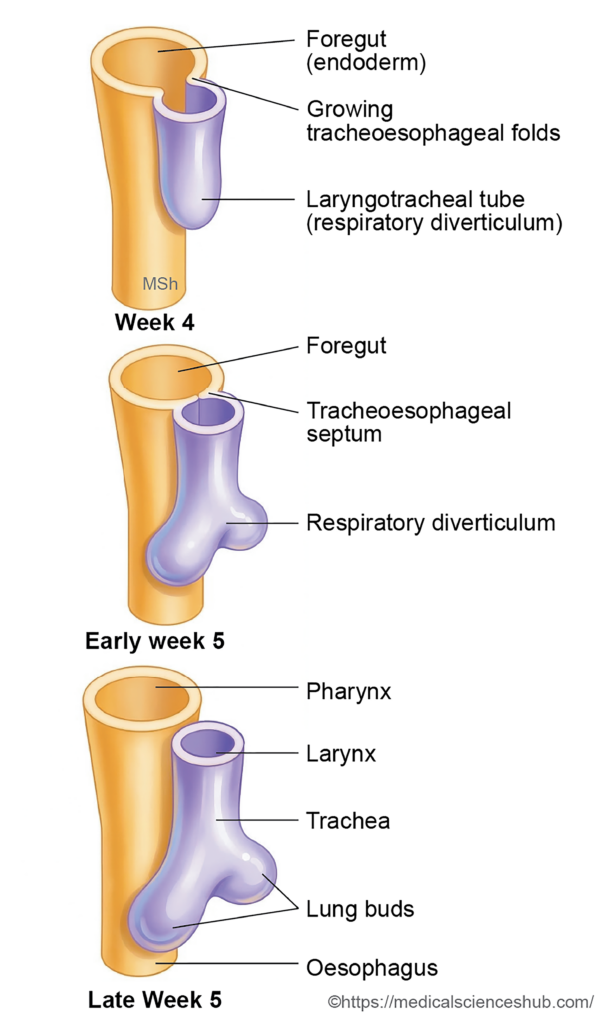
- During the fourth week of development, a median depression known as the laryngotracheal groove appears in the floor of the primitive pharynx, just caudal to the hypobranchial eminence. This groove gradually deepens to form the laryngotracheal tube, also called the respiratory diverticulum, which elongates in a caudal direction.
- At its distal end, the laryngotracheal tube divides into two outgrowths, forming the right and left lung buds. These buds represent the early primordia of the future lungs.
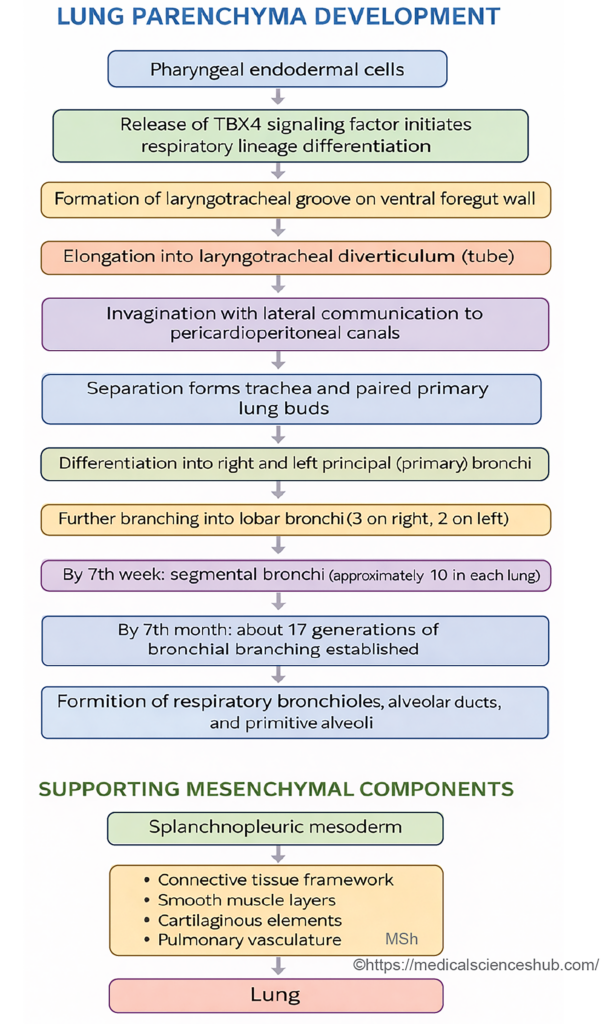
- Cells of the pharyngeal endoderm expressing the transcription factor TBX4 play an essential role in initiating the formation of the lung buds.
Fate of the Laryngotracheal Tube and Lung Buds
- The developing structures give rise to several components of the respiratory tract:
- The proximal portion of the laryngotracheal tube forms the larynx.
- The distal portion develops into the trachea.
- The lung buds differentiate into the bronchial tree and the lungs.
- The surrounding splanchnic mesoderm forms the supporting connective tissues, cartilage, smooth muscle, and vasculature of the respiratory system.
Separation of the Respiratory and Digestive Passages
- On each side of the laryngotracheal tube, two longitudinal ridges called the tracheoesophageal folds develop. These folds grow toward each other and fuse in the midline to form the tracheoesophageal septum, which separates the laryngotracheal tube anteriorly from the oesophagus posteriorly.
- The cranial extension of this septum is limited by the laryngeal inlet, also known as the furcula of His, which maintains communication between the larynx and the pharynx.
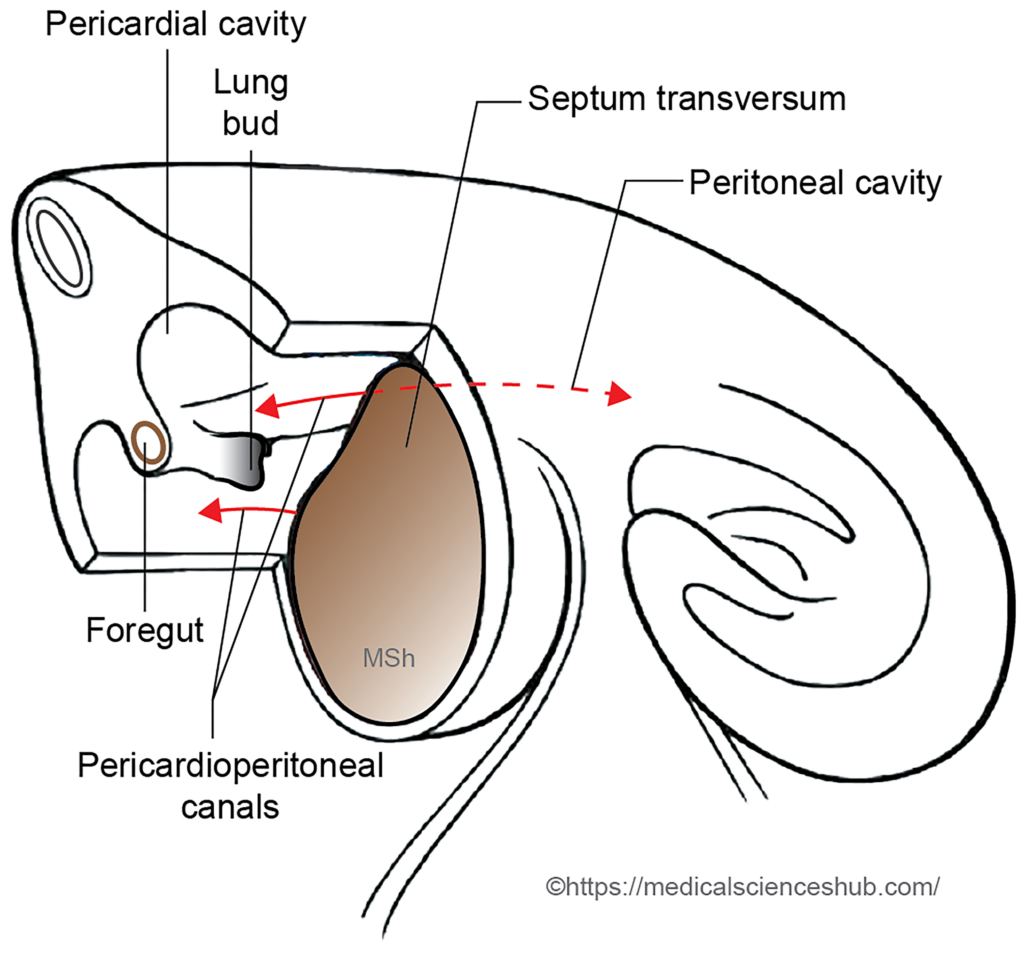
- The lung buds expand laterally and grow into the pericardioperitoneal canals, which subsequently develop into the pleural cavities.
DEVELOPMENT OF LARYNX
- The larynx (voice box) is a specialized component of the respiratory system that regulates airflow and produces sound. Structurally, it consists of the laryngeal inlet, vestibular folds, vocal folds, and supporting cartilages and muscles.
- The various components of the larynx originate from distinct embryological sources:
- Lining epithelium: The epithelial lining of the larynx develops from the endoderm of the laryngotracheal diverticulum, which arises from the foregut.
- Vestibular and vocal folds: The vestibular folds (false vocal cords) and vocal folds (true vocal cords) form from endodermal folds that develop within the laryngotracheal diverticulum.
- Laryngeal cartilages: Most laryngeal cartilages—thyroid, cricoid, arytenoid, corniculate, and cuneiform cartilages—are derived from the mesenchyme of the fourth and sixth pharyngeal arches. The epiglottis, however, develops from the hypobranchial eminence located in the floor of the primitive pharynx.
- Muscles of the larynx: Nearly all intrinsic laryngeal muscles originate from the sixth pharyngeal arch, while the cricothyroid muscle specifically develops from the fourth pharyngeal arch.
- Nerve supply: The embryological arch origin determines the nerve supply:
- The superior laryngeal nerve (branch of the vagus nerve) supplies structures derived from the fourth arch.
- The recurrent laryngeal nerve supplies derivatives of the sixth arch.
- This coordinated development of epithelial, cartilaginous, muscular, and neural elements forms the functional laryngeal apparatus responsible for phonation and airway protection.
Stages of Laryngeal Development
- The larynx develops from the cranial portion of the laryngotracheal diverticulum, an endodermal outgrowth arising from the floor of the primitive pharynx. The opening between the diverticulum and the pharynx persists as the laryngeal inlet.
- As development progresses, differential growth of the fourth and sixth pharyngeal arches modifies the surrounding mesenchyme. This process transforms the initial opening of the larynx into a characteristic T-shaped laryngeal orifice.
- The endodermal lining of the larynx undergoes rapid proliferation, temporarily occluding the laryngeal lumen. Subsequently, the lumen reopens through a process of recanalization. During this stage, the endoderm forms two pairs of folds: the superior vestibular folds and the inferior vocal folds. The vestibular folds later develop into the false vocal cords, whereas the vocal folds form the true vocal cords. The depression between these folds becomes the laryngeal ventricle.
- The mesenchyme of the fourth and sixth pharyngeal arches differentiates into most of the laryngeal cartilages, including the thyroid, cricoid, arytenoid, corniculate, and cuneiform cartilages. In contrast, the epiglottis arises from the hypobranchial eminence in the floor of the pharynx.
- The intrinsic muscles of the larynx originate primarily from the sixth pharyngeal arch, except for the cricothyroid muscle, which develops from the fourth arch. Consequently, most laryngeal muscles are supplied by the recurrent laryngeal nerve, whereas the cricothyroid muscle receives innervation from the external branch of the superior laryngeal nerve.
- The vocal folds mark the boundary between derivatives of the fourth and sixth arches. Accordingly, the mucosa above the vocal folds is supplied by the internal laryngeal branch of the vagus nerve (fourth arch derivative), while the mucosa below the vocal folds receives innervation from the recurrent laryngeal nerve (sixth arch derivative).
Table 15.1: Embryological Development of the Larynx
| Component | Embryological Origin & Key Details |
|---|---|
| Laryngeal mucosa | Derived from endoderm of proximal laryngotracheal tube arising from ventral foregut. |
| Vestibular & vocal folds | Form from endodermal folds of laryngotracheal tube; epithelial proliferation temporarily occludes lumen, followed by recanalization forming laryngeal ventricles and separating false and true folds. |
| Laryngeal cartilages | Develop mainly from mesenchyme of 4th and 6th pharyngeal arches; epiglottis arises from hypobranchial eminence. |
| Intrinsic muscles | Mostly derived from mesoderm of 6th arch; cricothyroid muscle originates from 4th arch. |
| Motor innervation | 6th arch muscles supplied by recurrent laryngeal nerve (CN X); cricothyroid (4th arch) by external branch of superior laryngeal nerve. |
| Sensory innervation | Above vocal folds: internal branch of superior laryngeal nerve; below vocal folds: recurrent laryngeal nerve. |
CLINICAL EMBRYOLOGY
Congenital Anomalies of the Larynx
- Laryngocele: A laryngocele is an abnormal air-filled dilation of the laryngeal saccule that communicates with the laryngeal lumen. It develops when the saccule becomes excessively enlarged and may extend either internally into the laryngeal cavity or externally through the thyrohyoid membrane into the neck. Clinically, it may present as a neck swelling that increases in size during coughing, straining, or phonation.
- Congenital Laryngeal Atresia and Stenosis: These abnormalities arise from failure of recanalization of the laryngeal lumen during embryonic development.
- Laryngeal atresia refers to complete obstruction of the laryngeal airway. It is a rare but severe anomaly that can result in life-threatening respiratory distress at birth.
- Laryngeal stenosis involves partial narrowing of the laryngeal lumen due to incomplete recanalization. The severity varies and may cause stridor, breathing difficulty, or voice changes.
- Laryngoptosis: Laryngoptosis describes an abnormal downward displacement of the larynx from its usual anatomical position. This condition may occur due to developmental defects in the supporting structures of the larynx, including abnormal formation or absence of certain laryngeal cartilages.
- Laryngeal Web: A laryngeal web is a thin membranous sheet that partially obstructs the laryngeal lumen, most commonly at the level of the vocal folds. It results from incomplete recanalization of the laryngeal cavity during embryonic development. The condition may lead to hoarseness, weak cry in infants, or airway obstruction depending on the extent of the membrane.
- Laryngomalacia: Laryngomalacia is the most common congenital anomaly of the larynx. It is characterized by flaccidity of the supraglottic structures, particularly the epiglottis and aryepiglottic folds. During inspiration, these structures collapse inward, partially obstructing the airway and producing inspiratory stridor in infants.
- Laryngeal Cysts: Congenital laryngeal cysts are fluid-filled sacs that develop from obstruction of mucus-secreting glands within the laryngeal mucosa. Depending on their size and location, these cysts may interfere with breathing, feeding, or phonation.
- Bifid Epiglottis: A bifid epiglottis is a rare developmental anomaly in which the epiglottis is split into two parts due to incomplete fusion during embryogenesis. This condition may occur in isolation or as part of certain congenital syndromes and may cause airway or swallowing difficulties.
DEVELOPMENT OF TRACHEA
- The trachea develops from the intermediate portion of the laryngotracheal tube, located between the developing larynx and the point where the tube divides to form the lung buds.
- The structural components of the trachea arise from different embryological layers:
- Lining epithelium and glands
The epithelial lining and associated tracheal glands originate from the endoderm of the laryngotracheal tube. - Supporting structures
The cartilage rings, connective tissue, and the trachealis muscle develop from the surrounding splanchnopleuric (splanchnic) mesoderm.
- Lining epithelium and glands
- During early development, the trachea and oesophagus share a common foregut cavity. They become separated when the tracheoesophageal folds fuse in the midline to form the tracheoesophageal septum, which divides the laryngotracheal tube anteriorly from the oesophagus posteriorly.
CLINICAL EMBRYOLOGY
Anomalies of Trachea
- Tracheal agenesis: It occurs when the laryngotracheal tube fails to develop, resulting in complete absence of the trachea.
- Tracheal stenosis: It refers to abnormal narrowing of the tracheal lumen. It may result from abnormal development or ventral displacement of the tracheoesophageal septum.
- Tracheal bronchus: In this anomaly, a small accessory bronchus or blind diverticulum arises from the trachea rather than from the main bronchi.
- Tracheal lobe: Occasionally, an accessory bronchus originates directly from the trachea and supplies a separate portion of lung tissue known as a tracheal lobe.
- Tracheoesophageal Fistula (TEF)
- Definition: Tracheoesophageal fistula (TEF) is a congenital abnormal communication between the trachea and the oesophagus. Although usually developmental, it may rarely occur as an acquired condition.
- Incidence: The condition occurs in approximately 1 in 2,500–4,000 live births.
- Embryological Basis: During normal development, the right and left tracheoesophageal folds fuse to form the tracheoesophageal septum, which separates the trachea anteriorly from the oesophagus posteriorly. Failure or abnormal fusion of these folds results in TEF.
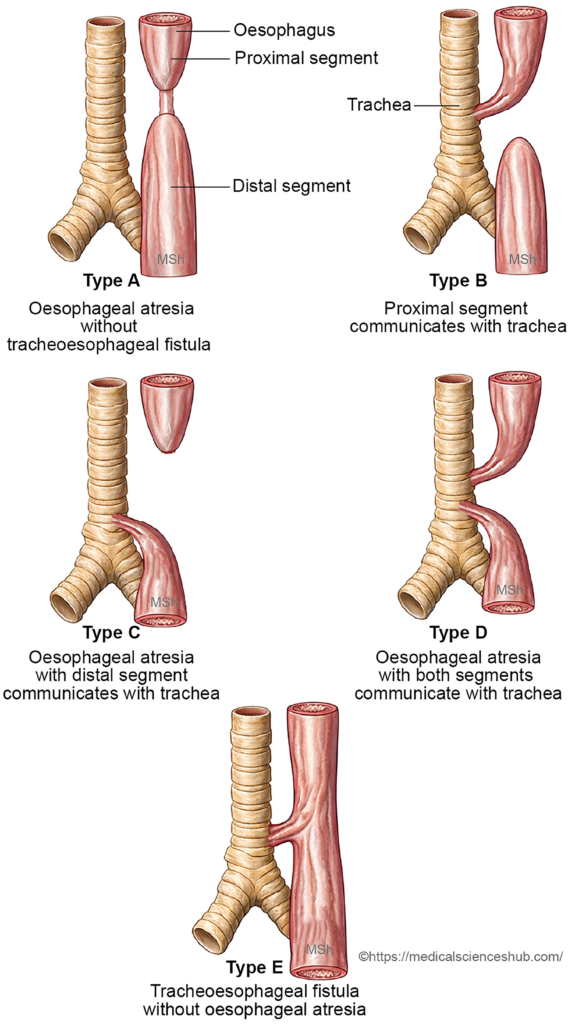
- Types of Tracheoesophageal Fistula
- TEF is classified according to the relationship between the trachea and oesophagus:
- Type A – Oesophageal atresia without fistula
The proximal and distal oesophageal segments end blindly and do not communicate with each other or with the trachea. - Type B – Proximal fistula with oesophageal atresia
The proximal oesophageal segment communicates with the trachea, while the distal segment ends as a blind pouch. - Type C – Distal fistula with oesophageal atresia
The proximal oesophagus forms a blind pouch, and the distal oesophagus communicates with the trachea. This is the most common form (about 85–90%). - Type D – Double fistula
Both the proximal and distal oesophageal segments communicate with the trachea. - Type E – Isolated tracheoesophageal fistula (H-type)
The trachea and oesophagus communicate through a fistulous tract, but no oesophageal atresia is present.
- Type A – Oesophageal atresia without fistula
- Clinical Features: In many cases, oesophageal atresia prevents the fetus from swallowing amniotic fluid, leading to polyhydramnios during pregnancy. After birth, affected infants typically develop coughing, choking, vomiting, and cyanosis during feeding because milk may enter the respiratory tract.
- Treatment: Management requires urgent surgical correction, which usually involves ligation of the fistula and anastomosis of the oesophageal segments to restore normal continuity of the oesophagus.
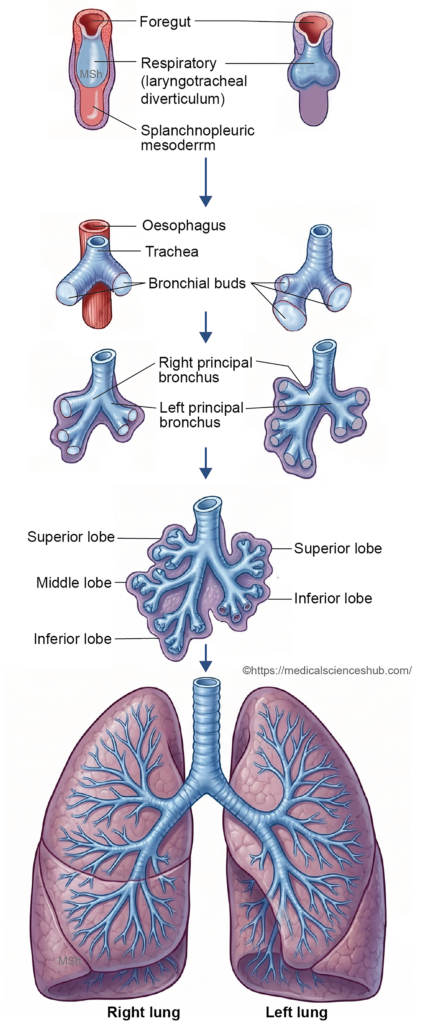
DEVELOPMENT OF BRONCHI AND LUNGS
- During the fifth week of intrauterine life, the laryngotracheal diverticulum divides into two branches that form the right and left principal bronchi. These early outgrowths, initially called lung buds, represent the primordia of the bronchial tree and lungs.
Formation of the Pleural Cavities and Pleura
- The lung buds grow rapidly in caudal and lateral directions, projecting into the pleuroperitoneal canals. As development progresses, the enlarging lungs gradually fill these canals. Subsequently, the canals expand further to accommodate continued lung growth.
- Two embryonic membranes help partition the developing thoracic cavity. The pleuropericardial folds separate the pleural cavities from the pericardial cavity, while the pleuroperitoneal folds divide them from the peritoneal cavity. After these partitions form, each pleuroperitoneal canal becomes an independent pleural cavity.
- The pleural membranes originate from mesodermal layers surrounding the lung buds. The splanchnopleuric mesoderm, which directly covers the developing lungs, forms the visceral pleura, whereas the somatopleuric mesoderm lining the thoracic wall gives rise to the parietal pleura.
Formation of Intrapulmonary Bronchi
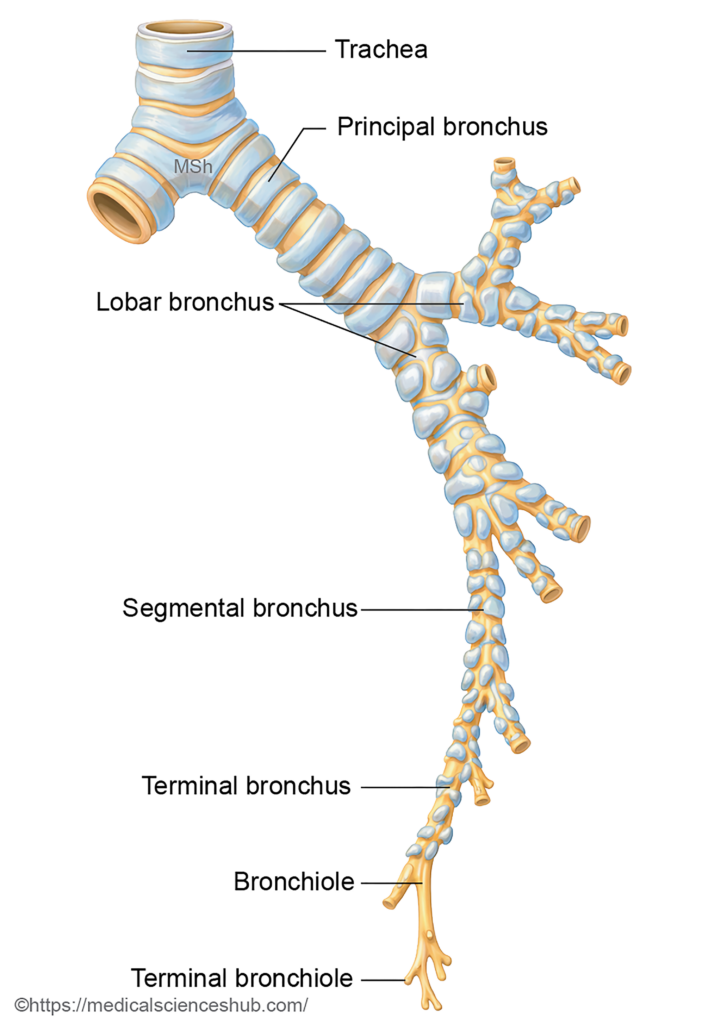
- The principal bronchi undergo successive branching within the developing lungs to form the intrapulmonary bronchial tree.
- The left principal bronchus divides into upper and lower secondary (lobar) bronchi, while the right principal bronchus forms superior, middle, and inferior lobar bronchi. Each secondary bronchus supplies a specific lung lobe, which later becomes separated by interlobar fissures.
- Around the seventh week of intrauterine life, the lobar bronchi further divide to produce segmental bronchi. Each segmental bronchus, together with surrounding splanchnopleuric mesoderm, gives rise to a bronchopulmonary segment, the structural and functional unit of the lung.
- By the end of the seventh month of gestation, approximately 17 generations of bronchial branching have formed. After birth, an additional 6–7 divisions occur before the bronchial tree reaches the adult pattern.
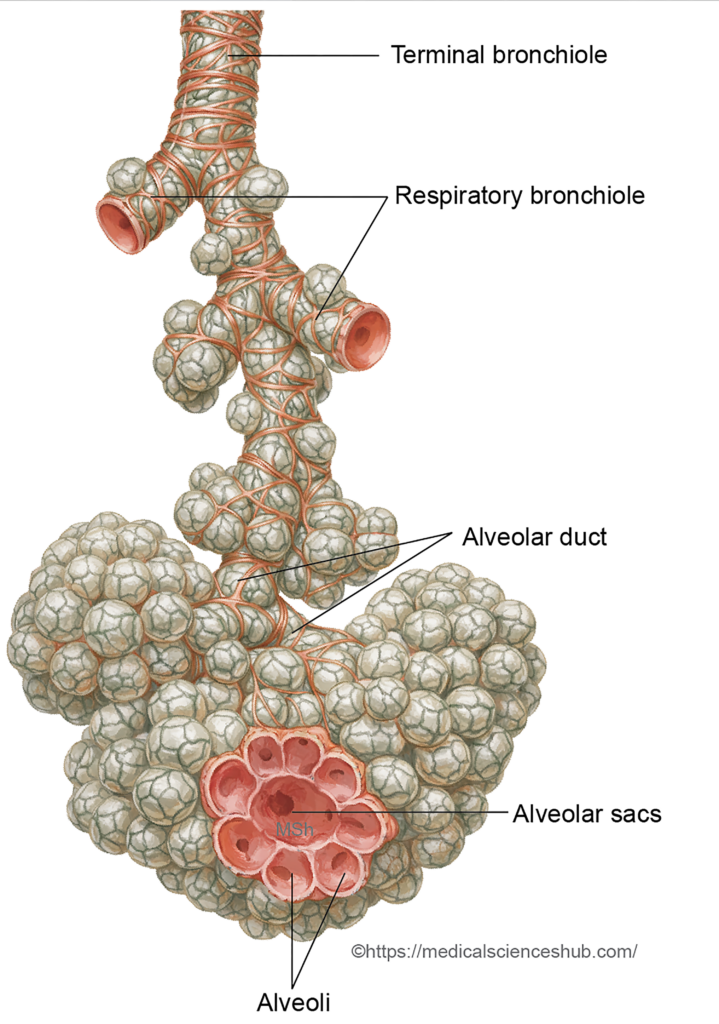
- The distal branches differentiate into bronchioles, respiratory bronchioles, alveolar ducts, and eventually alveoli.
- Before birth, each lung develops approximately 20–70 million terminal sacs. Postnatal growth increases the number of alveoli to about 300–400 million in the mature lungs, greatly expanding the surface area for gas exchange.
Parenchyma of the Lung
- The epithelial lining of the bronchial tree and alveoli is derived from the endoderm of the respiratory diverticulum. In contrast, the cartilage, blood vessels, smooth muscle, and connective tissue components of the lung develop from the surrounding splanchnopleuric mesoderm.
Maturation of the Lungs
The development and functional maturation of the lungs occur through a sequence of well-defined stages during fetal and early postnatal life. These stages reflect progressive differentiation of the bronchial tree, terminal air spaces, and structures required for gas exchange.
Lung maturation is commonly divided into four developmental periods:
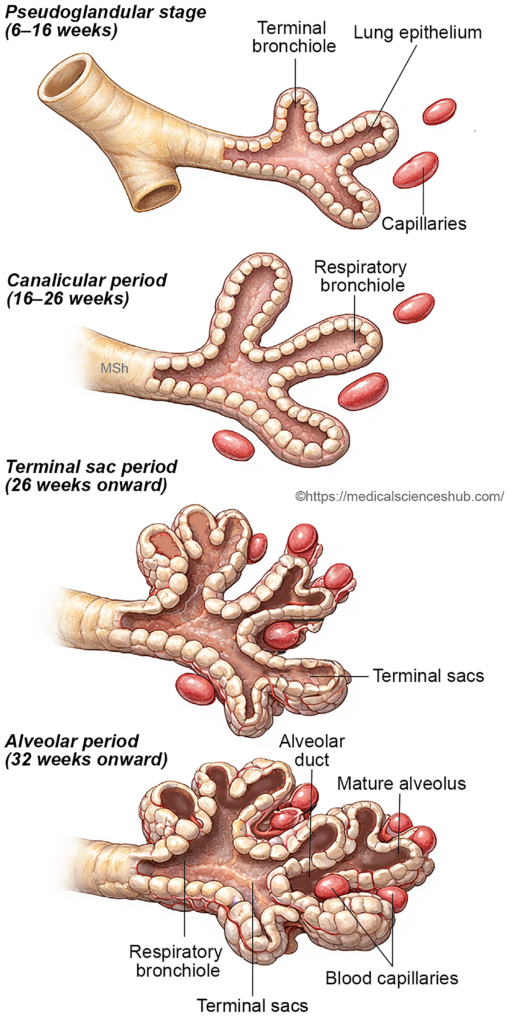
- Pseudoglandular period (6–16 weeks): During this stage, extensive branching of the bronchial tree occurs, forming terminal bronchioles. The lung tissue resembles a glandular structure, and functional respiratory units have not yet developed.
- Canalicular period (16–26 weeks): The lung tissue becomes more vascular, and the lumina of bronchioles enlarge. Differentiation of respiratory bronchioles and early alveolar ducts begins, marking the initial development of structures capable of limited gas exchange.
- Terminal saccular period (26 weeks to birth): Numerous terminal sacs (primitive alveoli) form, and the epithelial lining becomes thinner. Type I and Type II pneumocytes differentiate, and surfactant production begins, improving the lung’s ability to function after birth.
- Alveolar period (approximately 32 weeks to about 8 years): Mature alveoli develop from the terminal sacs. Their number increases markedly after birth through continued alveolar septation, allowing progressive expansion of the respiratory surface area.
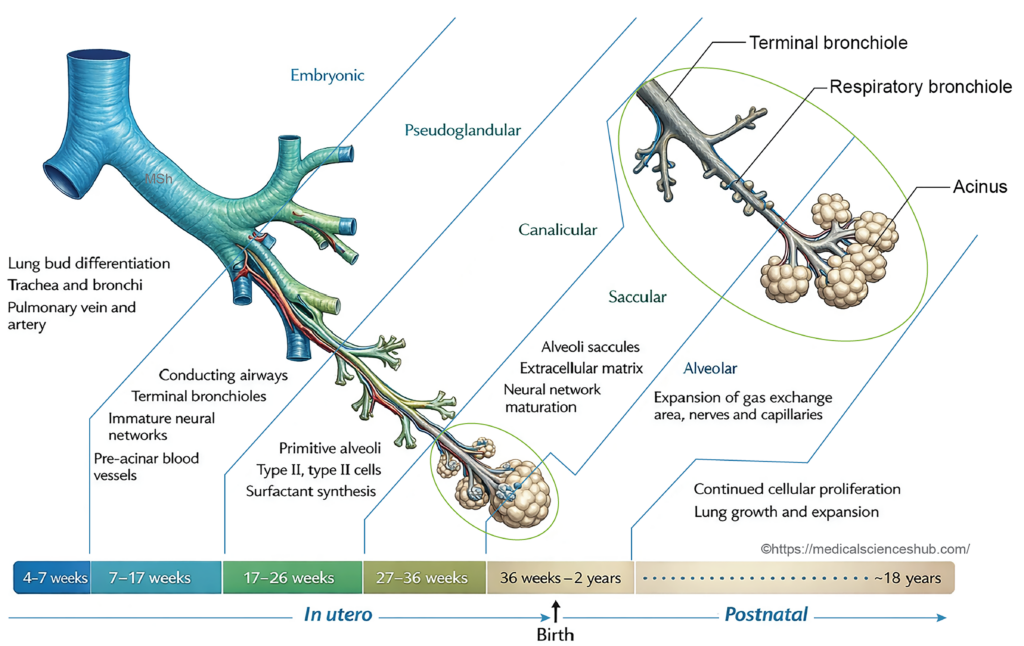
Table 15.2: Stages of Lung Maturation
| Stage (Time Period) | Key Morphological & Functional Features |
|---|---|
| Pseudoglandular (6–16 weeks) | Lung appears gland-like Exextensive branching forms bronchi, bronchioles, and terminal bronchioles. No respiratory bronchioles or alveoli No gas exchange possible, so survival outside uterus is not feasible. |
| Canalicular (16–26 weeks) | Lumina enlarge and vascularity increases. Respiratory bronchioles, alveolar ducts, and primitive alveoli begin forming. Type I and II pneumocytes differentiate. Limited survival possible near end with intensive care. |
| Terminal Sac (26 weeks–birth) | Numerous terminal sacs develop. Blood–air barrier thins with close capillary–epithelial contact. Type I cells predominate; type II cells increase. Surfactant production rises, improving lung stability. |
| Alveolar (32 weeks–~8 years) | Mature alveoli form and increase via septation. Postnatal growth expands surface area and capillary networks. Continued surfactant production supports respiration; near-adult alveolar number reached by ~8 years. |
CLINICAL EMBRYOLOGY
- Pulmonary Agenesis and Hypoplasia: Pulmonary agenesis: Complete absence of lung, bronchus, and pulmonary vessels due to failure of development of the primary bronchial bud.
- Pulmonary hypoplasia: Incomplete lung development with reduced bronchi, alveoli, and pulmonary vessels. Often associated with oligohydramnios or congenital diaphragmatic hernia.
- Abnormal Lobulation of the Lung: Results from abnormal formation of lung fissures.
- Incomplete or absent fissures: Lobes are fused.
- Accessory fissures: Extra lobes may be formed.
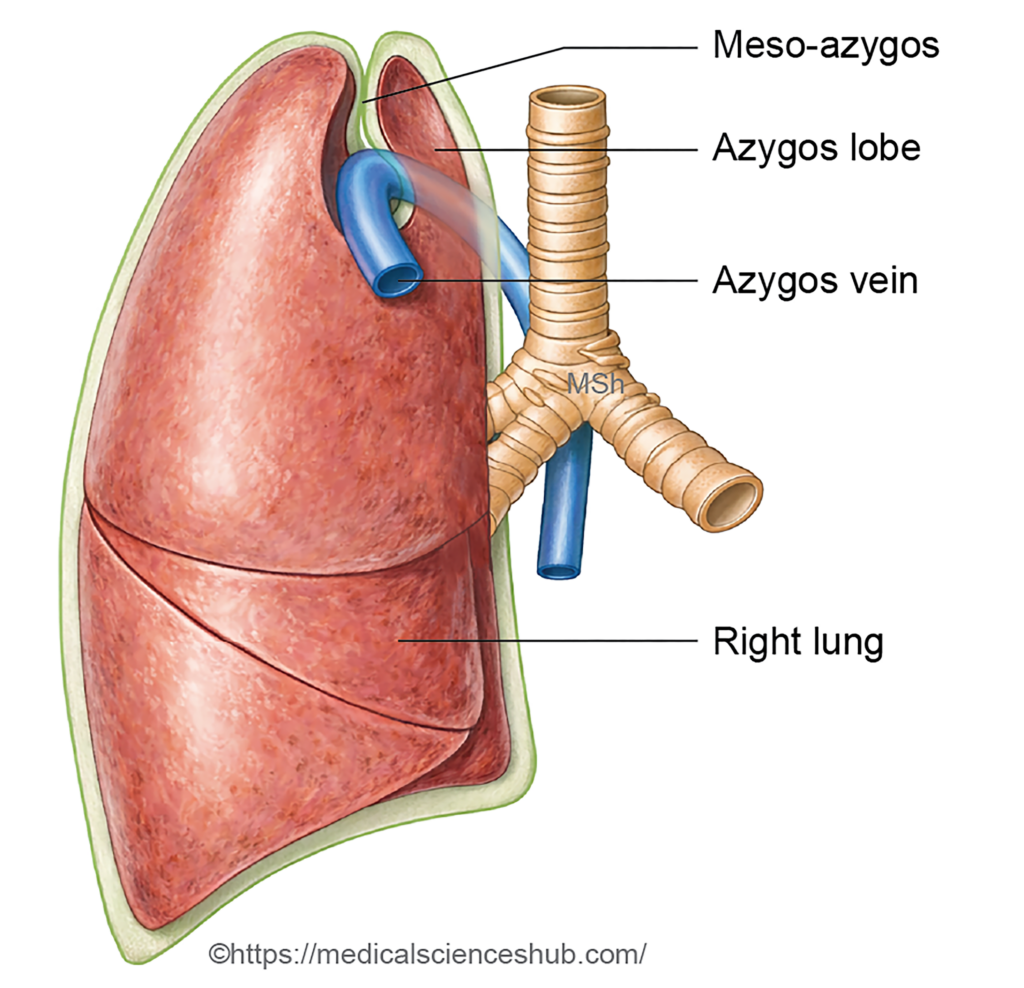
- Azygos Lobe (Lobe of Wrisberg): An accessory lobe of the right lung formed when the azygos vein penetrates the upper lobe during development, creating an accessory fissure. Usually asymptomatic.
- Pulmonary Sequestration: A mass of lung tissue that does not communicate with the bronchial tree and receives arterial supply from systemic circulation (usually the aorta). It may be intralobar or extralobar.
- Ectopic Lung Tissue: Lung tissue located outside the normal lung due to abnormal separation of the respiratory diverticulum during early development.
- 6. Congenital Polycystic Lung (Congenital Pulmonary Airway Malformation – CPAM): Developmental anomaly characterized by multiple cysts formed by abnormal proliferation of terminal bronchioles. It may cause respiratory distress in newborns.
- 7. Bronchogenic Cysts: Congenital cysts formed by abnormal budding of the tracheobronchial tree. Commonly located in the mediastinum or lung.
- Congenital Lobar Emphysema: Overdistension of a pulmonary lobe caused by defective bronchial cartilage leading to airway obstruction and air trapping, usually affecting the left upper lobe or right middle lobe.
Hyaline membrane disease or infant respiratory distress syndrome (IRDS)
- Hyaline membrane disease, also called infant respiratory distress syndrome (IRDS), is a serious respiratory disorder that primarily affects premature infants. The condition results from inadequate production of pulmonary surfactant by type II pneumocytes in the developing lungs.
- Surfactant reduces alveolar surface tension and helps keep the alveoli open during breathing. When surfactant levels are insufficient, the alveoli tend to collapse during expiration (atelectasis). This leads to impaired gas exchange and progressive respiratory difficulty shortly after birth.
- The disorder is closely related to the immaturity of the lungs, particularly when birth occurs before the stage at which surfactant production becomes adequate. As a consequence of alveolar collapse and epithelial injury, a protein-rich exudate accumulates along the alveolar walls, forming characteristic hyaline membranes.
- Clinically, affected newborns develop rapid breathing, cyanosis, and respiratory distress soon after delivery. Management typically includes respiratory support and administration of exogenous surfactant, which significantly improves survival and lung function.
Important Questions
- Describe the embryological development of the larynx.
- Explain the embryological basis, formation, and clinical significance of tracheoesophageal fistula.
- Outline the embryological development of the lungs.
- Discuss the stages involved in the maturation of the lungs during fetal development.
- What is an azygos lobe? Describe its developmental basis and anatomical significance.
- Write a short note on hyaline membrane disease, including its embryological basis and clinical features.

