Competencies
- AN52.6: Describe the development and congenital anomalies of foregut, midut and hindgut.
Note: Liver, gallbladder, pancreas and spleen develop in relation to gut tube. Gastrointestinal tract develops to form foregut, midgut and hindgut. The liver, gallbladder, pancreas and spleen develop in relation to the foregut and midgut. This chapter includes development of liver, gallbladder, pancreas, and spleen.
DEVELOPMENT OF LIVER
The liver originates from multiple embryonic sources that contribute to its parenchyma, supporting tissues, and vascular structures.
- The hepatic bud (also called the hepatic diverticulum), which arises from the endoderm of the foregut, gives rise to the hepatocytes and the intrahepatic biliary system, including the bile ducts within the liver.
- The septum transversum, a mass of mesodermal tissue, contributes to the connective tissue framework of the liver. It forms the fibrous capsule, stromal components, and provides the source of Kupffer cells and part of the hepatic vascular connective tissue.
- The vitelline veins and umbilical veins are incorporated into the developing liver and differentiate to form the hepatic sinusoids, which establish the characteristic vascular channels of the liver.
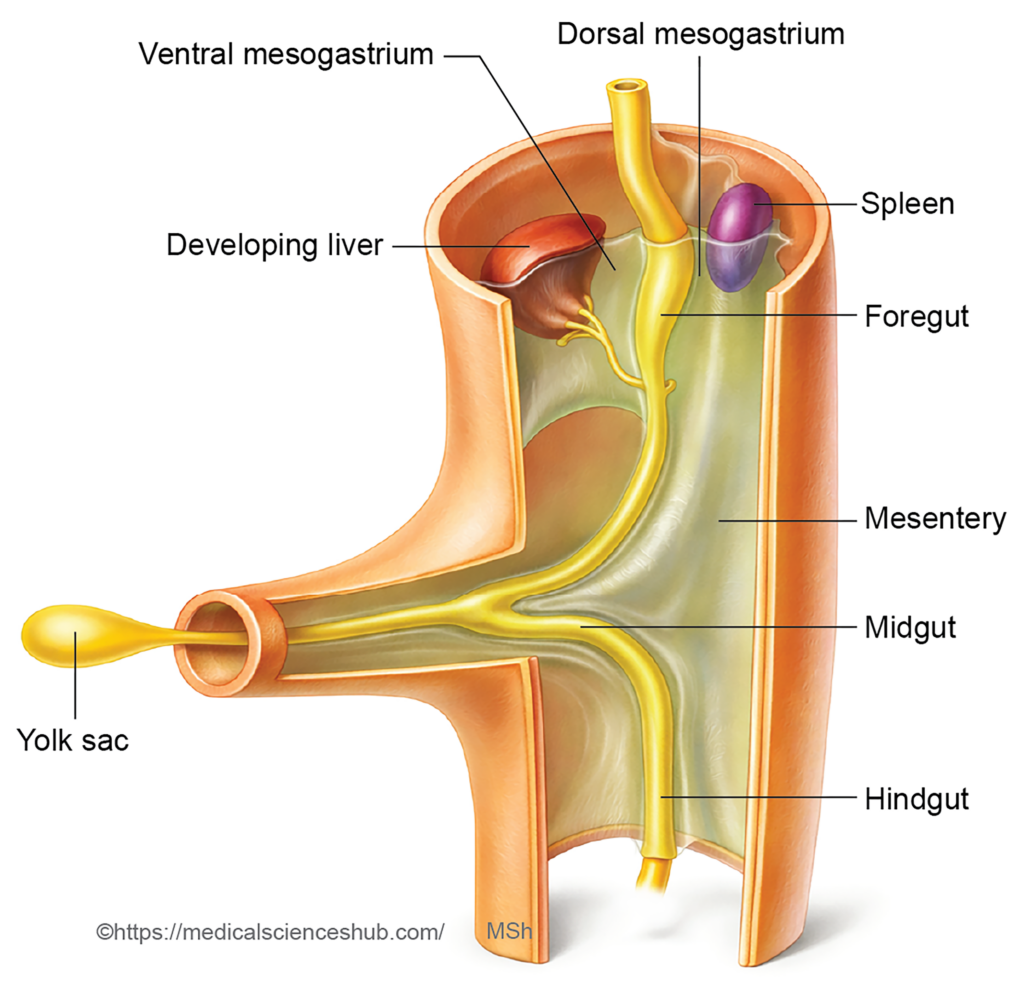
- The ventral mesentery contributes to the formation of several peritoneal ligaments associated with the liver, including the lesser omentum, falciform ligament, coronary ligament, and triangular ligaments.
Stages of Development
- Formation of the hepatic bud
During the third week of intrauterine life, a small outgrowth known as the hepatic bud (hepatic diverticulum) emerges from the ventral wall of the foregut, near the region that later forms the second part of the duodenum. - Growth and cellular differentiation
The hepatic bud enlarges and extends cranially and ventrally into the ventral mesentery, reaching the septum transversum. It produces branching cords of primitive liver cells called hepatoblasts. These cells later differentiate into hepatocytes, form the bile canaliculi, and contribute to the developing hepatic ducts. - Division of the hepatic bud
The bud divides into a larger pars hepatica and a smaller pars cystica. The pars hepatica forms the liver parenchyma and gives rise to the right and left hepatic ducts, while the pars cystica develops into the gallbladder and cystic duct. Cells of the hepatic ducts organize into interlacing columns called hepatic trabeculae. - Development of intrahepatic biliary passages
Branching of the hepatic ducts produces the intrahepatic biliary channels, which later establish connections with the bile canaliculi within the liver tissue. - Formation of hepatic sinusoids
As the hepatic trabeculae expand and rearrange, spaces appear between them that become hepatic sinusoids. The vitelline and umbilical veins, which traverse the septum transversum, fragment and integrate with these sinusoids to establish the liver’s vascular network. Evidence suggests that a portion of the endothelial cells lining the sinusoids, portal veins, and central veins may originate from the sinus venosus. - Formation of liver capsule and stromal components
Mesenchymal tissue from the septum transversum forms the connective tissue stroma of the liver, including the fibrous capsule, blood vessels, Kupffer cells, and early hematopoietic cells. - Development of peritoneal ligaments
As the liver enlarges, it divides the ventral mesentery into the falciform ligament and lesser omentum. Peritoneal reflections between the liver and diaphragm form the coronary and triangular ligaments. Most of the liver becomes covered by visceral peritoneum, except for a superior region that remains attached to the septum transversum; this uncovered area is known as the bare area of the liver.
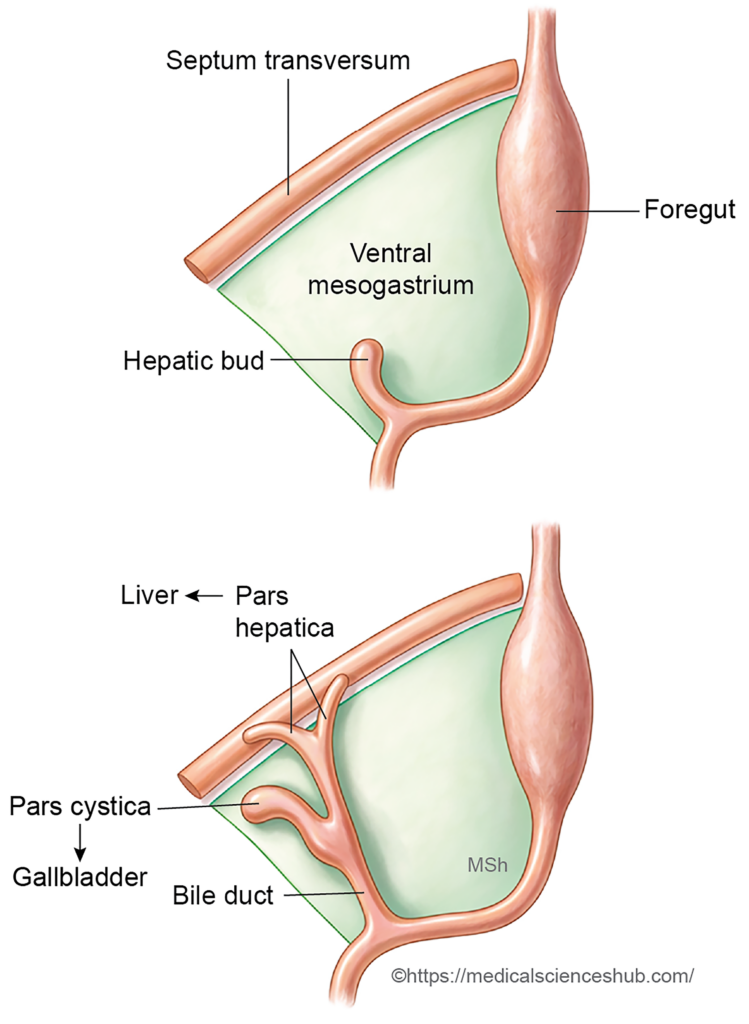
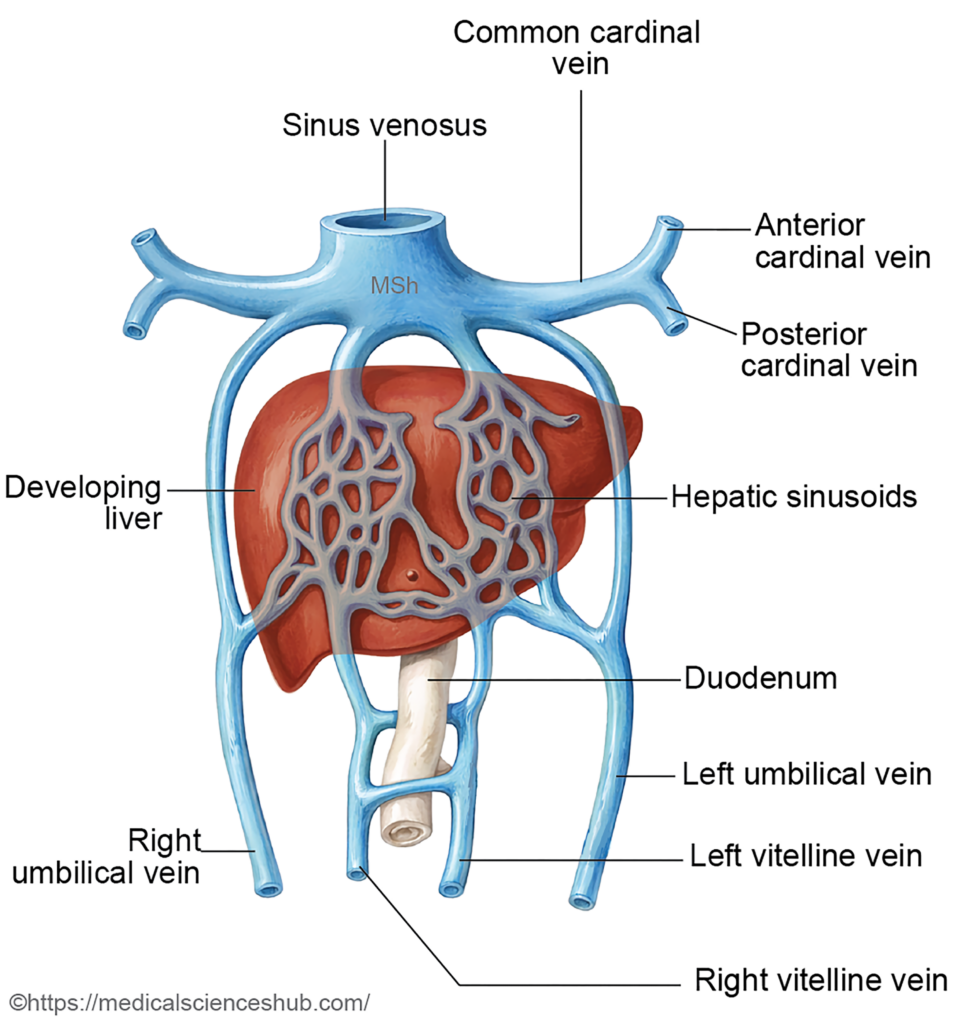
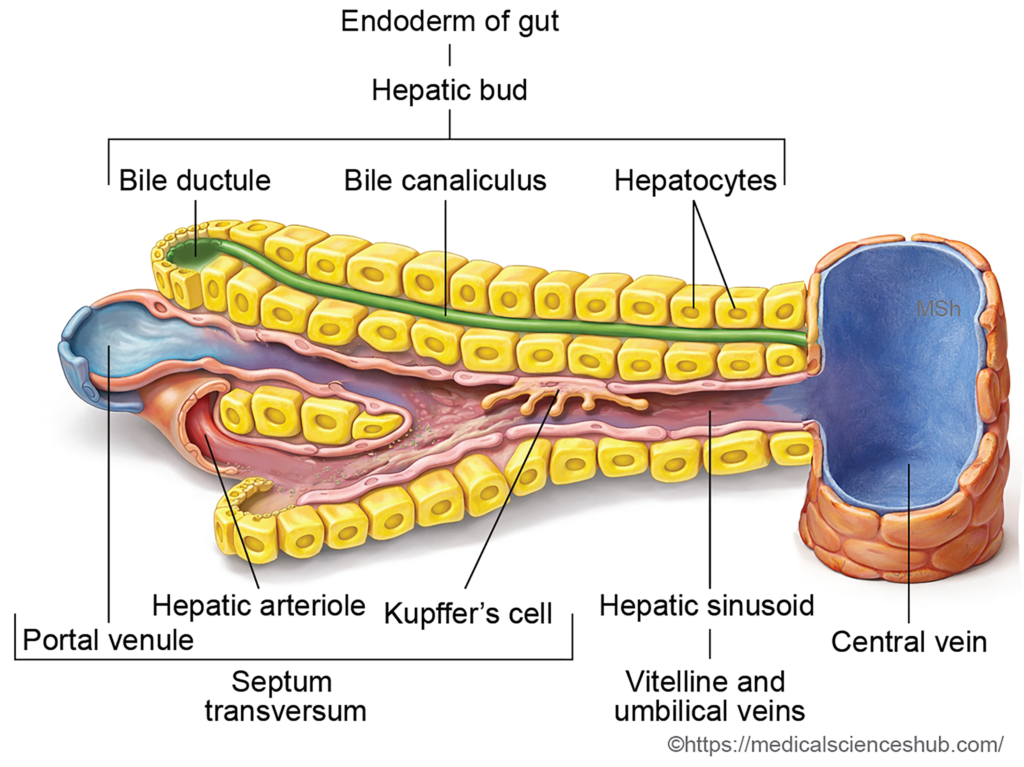
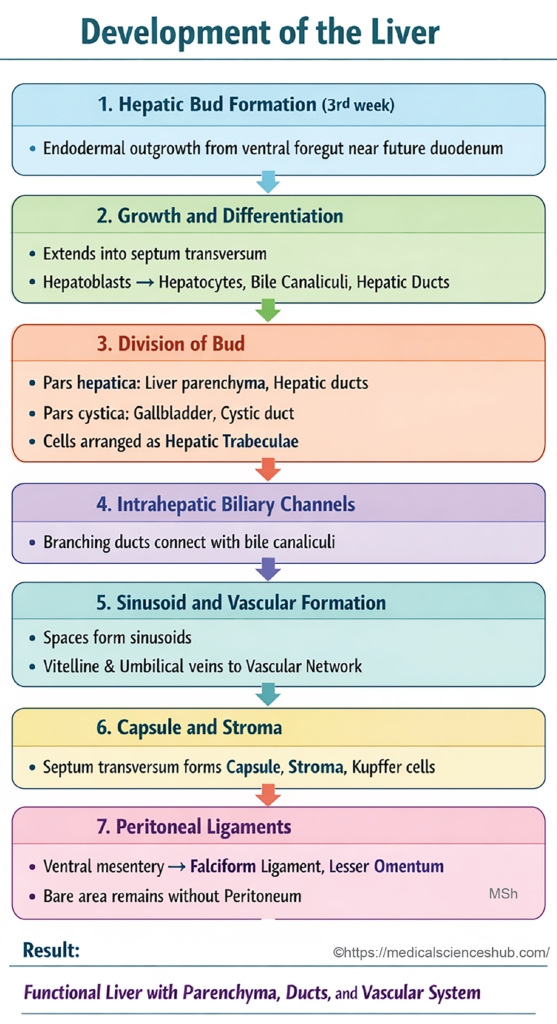
Table 14.1: Development of liver
| Component/Structure | Origin and Development |
|---|---|
| Hepatic tissue (lobes) | Derived from endoderm via the hepatic diverticulum from foregut (duodenum). Forms the bulk of liver. |
| Hepatocytes | Differentiate from proliferating endodermal cells of the hepatic bud invading surrounding mesenchyme. |
| Bile canaliculi | Develop as intercellular channels between adjacent hepatocytes. |
| Intrahepatic bile ducts | Form from branching epithelial cords of the hepatic diverticulum. |
| Connective tissue framework | Arises from mesoderm of septum transversum, providing structural support. |
| Glisson’s capsule | Fibrous covering derived from septum transversum mesoderm. |
| Stroma | Internal connective tissue from mesodermal mesenchyme. |
| Kupffer cells | Originate from mesodermal hematopoietic cells; function as macrophages. |
| Hepatic vessels | Develop within mesoderm associated with septum transversum. |
| Liver sinusoids | Form by remodeling of vitelline and umbilical veins. |
| Peritoneal ligaments | Derived from ventral mesogastrium. |
| • Falciform ligament | Connects liver to anterior abdominal wall. |
| • Lesser omentum | Connects liver to stomach and duodenum. |
| • Coronary & triangular ligaments | Anchor liver to diaphragm. |
| Ligamentum teres | Remnant of left umbilical vein. |
| Ligamentum venosum | Remnant of ductus venosus (fetal shunt). |
CLINICAL EMBRYOLOGY
Congenital Anomalies of the Liver
- Riedel’s lobe: It is a downward, tongue-like projection from the right lobe of the liver. Because of its elongated shape, it may be mistaken clinically for an abdominal mass, sometimes leading to unnecessary diagnostic procedures or surgery.
- Polycystic liver disease (PLD): It is an uncommon disorder caused by abnormal development of the intrahepatic bile ducts, particularly defective connection between biliary ductules and larger bile ducts. This results in multiple hepatic cysts. The condition is often associated with polycystic kidney disease and occasionally with cysts in the pancreas. It is usually inherited as an autosomal dominant disorder.
- Intrahepatic biliary atresia: It results from failure of formation or obliteration of the intrahepatic bile ducts. This severe condition leads to impaired bile drainage and progressive liver damage, and in advanced cases liver transplantation may be required.
- Caroli disease: It is characterized by congenital cystic dilatation (ectasia) of the intrahepatic biliary tree, which predisposes to bile stasis, recurrent infection, and stone formation.
- Congenital hepatic fibrosis: It is an inherited fibrocystic liver disorder marked by excessive fibrous tissue deposition within the liver. Progressive fibrosis can obstruct portal blood flow and produce portal hypertension.
- Lobar and positional anomalies of the liver: Developmental variations may include absence of a hepatic lobe, the presence of an accessory lobe, or a rudimentary liver. In some cases, small islands of ectopic liver tissue may occur in structures such as the lesser omentum or falciform ligament.
DEVELOPMENT OF GALLBLADDER AND EXTRAHEPATIC BILIARY APPARATUS
Stages of Development
- The extrahepatic biliary apparatus develops primarily from endoderm derived from the embryonic foregut.
- During early development, the hepatic bud emerges as a ventral outgrowth from the terminal part of the foregut, which later contributes to the formation of the second part of the duodenum. This hepatic diverticulum grows ventrally and cranially into the ventral mesogastrium.
- As development progresses, the hepatic bud divides into two components: the pars hepatica and the pars cystica. The pars hepatica forms the liver parenchyma and gives rise to the right and left hepatic ducts. The pars cystica, also known as the cystic bud, enlarges and differentiates into the gallbladder and the cystic duct.
- The right and left hepatic ducts unite to form the common hepatic duct. This duct subsequently joins the cystic duct, producing the common bile duct.
- Initially, the common bile duct opens on the ventral surface of the developing duodenum. However, due to differential growth and rotation of the duodenal wall, the opening gradually shifts to the dorsomedial aspect of the duodenum. This positional change occurs together with the movement of the ventral pancreatic bud, reflecting coordinated development of the hepatobiliary and pancreatic systems.
Table 14.2: Development of extrahepatic biliary apparatus
| Structure | Origin | Development |
|---|---|---|
| Gallbladder | Cystic bud (pars cystica) | Enlarges and differentiates into a bile-storing organ. |
| Cystic duct | Cystic bud | Proximal narrow part persists as duct connecting gallbladder to biliary tree. |
| Right & left hepatic ducts | Pars hepatica (hepatic bud) | Branches form ducts draining bile from liver lobes. |
| Common bile duct | Proximal hepatic diverticulum | Forms main channel that joins cystic duct and opens into duodenum. |
CLINICAL EMBRYOLOGY
Anomalies of Gallbladder
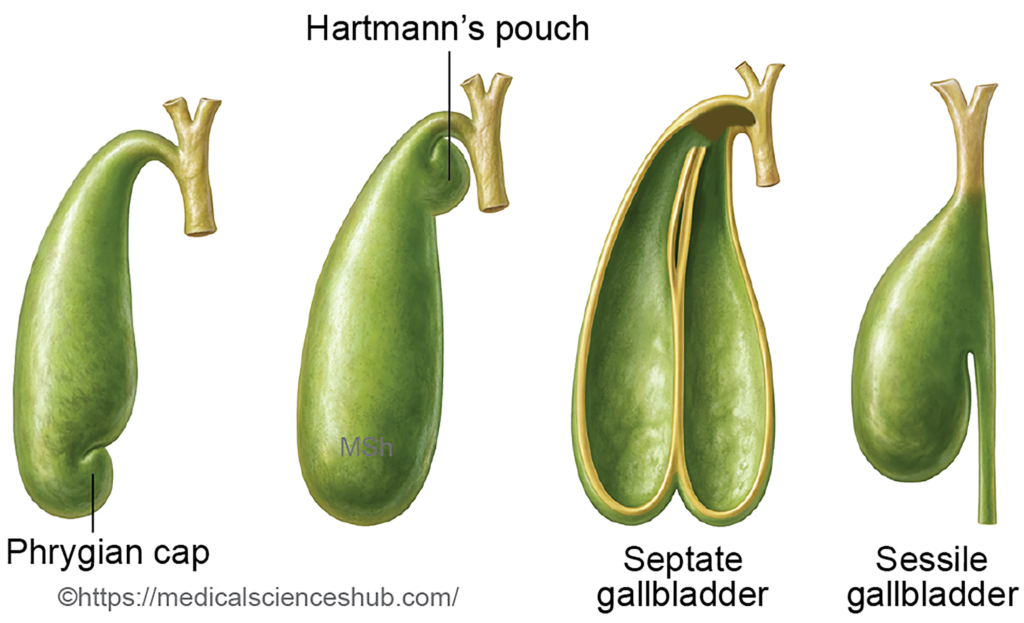
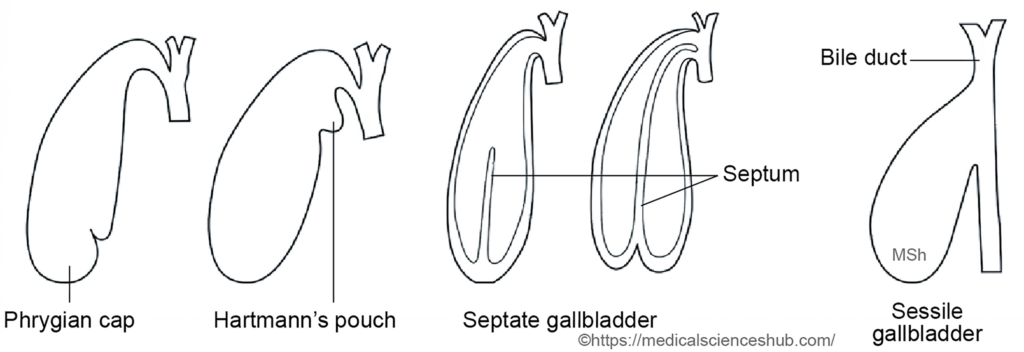
- Agenesis of the gallbladder: A rare congenital condition characterized by complete absence of the gallbladder due to failure of its embryonic development.
- Sessile gallbladder: This anomaly involves absence of the cystic duct, causing the gallbladder to open directly into the common bile duct.
- Phrygian cap deformity: A common anatomical variant in which the fundus of the gallbladder folds back upon itself, forming a cap-like projection.
- Hartmann’s pouch: A localized outpouching of the gallbladder neck, which may predispose to bile stasis and gallstone formation.
- Septate gallbladder: The lumen of the gallbladder is partially divided by internal septa, producing multiple communicating compartments.
- Double gallbladder: A congenital duplication in which two gallbladders are present. These may share a common cystic duct or possess separate ducts that join the biliary tree.
- Intrahepatic gallbladder: The gallbladder remains partially or completely embedded within the liver parenchyma rather than lying on its inferior surface.
- Floating gallbladder: The gallbladder is completely covered by peritoneum and attached to the liver by a mesentery, making it unusually mobile.
Anomalies of the Extrahepatic Biliary Ducts
- Biliary duct atresia: Partial or complete absence or obstruction of components of the extrahepatic biliary ducts, including the common bile duct, common hepatic duct, or hepatic ducts.
- Accessory bile ducts: Additional small biliary ducts may connect the liver directly to the gallbladder or to the extrahepatic biliary tree. These ducts are clinically important because they may be injured during hepatobiliary surgery.
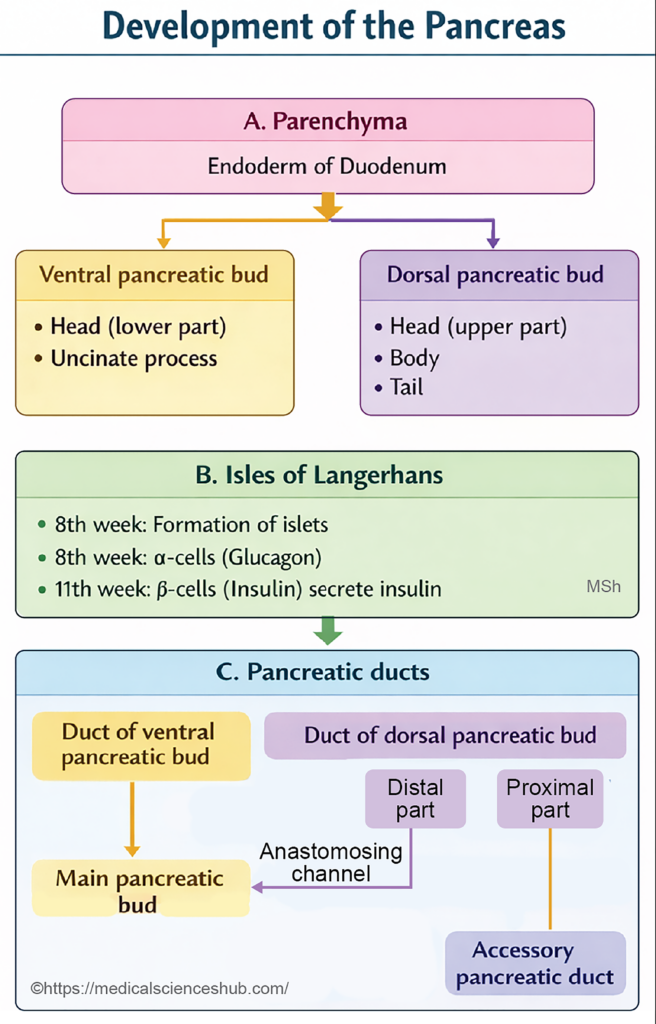
DEVELOPMENT OF PANCREAS
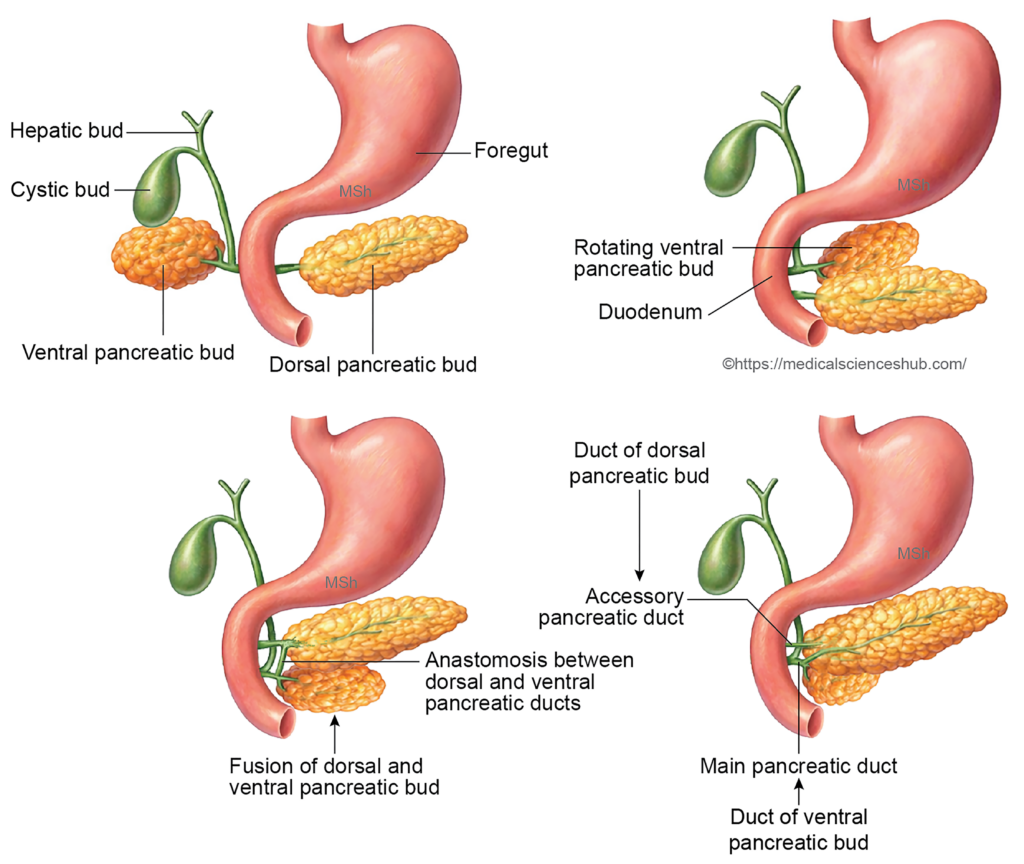
- The pancreas develops from two endodermal pancreatic buds, the dorsal pancreatic bud and the ventral pancreatic bud, which arise from the caudal region of the foregut near the foregut–midgut junction.
- Proliferation and branching of these endodermal buds give rise to the pancreatic duct system, the exocrine acinar cells, and the endocrine islets of Langerhans.
- The surrounding splanchnic mesoderm differentiates to form the connective tissue stroma, including the pancreatic capsule, blood vessels, and supporting structures.
Stages of Development of Pancreas
- Formation of pancreatic buds:
During the third week of development, two pancreatic buds arise from the terminal part of the foregut. The dorsal pancreatic bud develops earlier from the dorsal wall, while the ventral pancreatic bud emerges from the ventral wall in close association with the hepatic diverticulum. Both share a common opening into the duodenum. The ventral bud is smaller and positioned slightly distal to the dorsal bud. It grows within the ventral mesentery of the duodenum, whereas the dorsal bud extends into the dorsal mesentery. - Rotation and fusion of buds:
As the duodenum rotates and grows asymmetrically, the ventral pancreatic bud, together with the common bile duct, moves posteriorly and to the right side of the duodenum. It gradually approaches the dorsal pancreatic bud, and the two buds fuse by the seventh week of intrauterine life. - Formation of pancreatic ducts:
After fusion, the ducts of the two buds anastomose to form the pancreatic ductal system.- Main pancreatic duct (duct of Wirsung): Formed mainly from the ventral pancreatic duct, the distal part of the dorsal duct, and their connecting channel.
- Accessory pancreatic duct (duct of Santorini): Derived from the proximal portion of the dorsal pancreatic duct.
- Development of acini and islets:
Extensive branching of both pancreatic buds produces the exocrine pancreatic acini. Some cells detach from the developing ducts and differentiate into the endocrine islets of Langerhans, which begin insulin secretion around the tenth week of development. - Contribution of mesoderm:
The surrounding splanchnic mesoderm forms the capsule, connective tissue septa, and vascular supply of the pancreas. - Final anatomical position:
With the rotation of the duodenum, most of the pancreas becomes secondarily retroperitoneal, except the tail of the pancreas, which remains intraperitoneal within the lienorenal ligament.
Development of pancreas
| Structure/Component | SpermatozOrigin and Developmentoon |
|---|---|
| Inferior head & uncinate process | From ventral pancreatic bud; rotates posteriorly with duodenum and fuses with dorsal bud. |
| Superior head, neck, body & tail | From dorsal pancreatic bud; forms most of pancreatic tissue. |
| Main pancreatic duct (Wirsung) | Formed by ventral duct and distal part of dorsal duct after fusion. |
| Accessory duct (Santorini) | From proximal dorsal duct; may persist, opening at minor duodenal papilla. |
| Stroma & connective tissue | Derived from mesodermal mesenchyme, forming supporting framework and vessels. |

CLINICAL EMBRYOLOGY
Anomalies of Pancreas
- Pancreatic divisum:
This condition results from failure of fusion between the dorsal pancreatic bud and ventral pancreatic bud. Consequently, most pancreatic secretions drain through the accessory pancreatic duct into the minor duodenal papilla. It is the most common congenital anomaly of the pancreas. - Accessory (ectopic) pancreatic tissue:
Small deposits of ectopic pancreatic tissue may occur outside the pancreas, commonly in the wall of the stomach, duodenum, gallbladder, or Meckel’s diverticulum. The stomach is the most frequent location. - Inversion of pancreatic ducts:
In this rare variation, the main pancreatic duct develops from the dorsal pancreatic duct and opens into the minor duodenal papilla. The ventral pancreatic duct joins the common bile duct and opens at the major duodenal papilla.
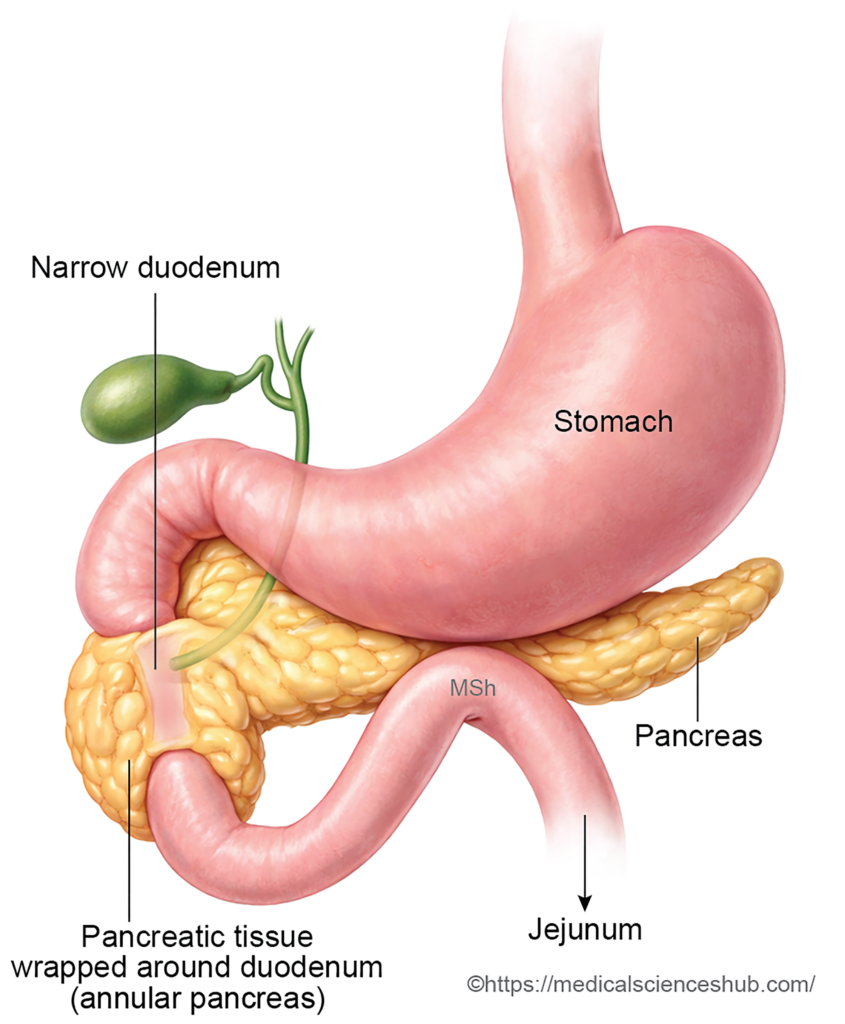
Annular Pancreas
- Annular pancreas is a congenital condition in which a ring of pancreatic tissue encircles the second part of the duodenum, potentially causing narrowing of the intestinal lumen.
- Incidence: Approximately 1 in 12,000–15,000 newborns, with a slightly higher occurrence in males.
- Cause: Normally, the ventral pancreatic bud rotates posteriorly around the duodenum and fuses with the dorsal pancreatic bud. Annular pancreas usually develops when the ventral bud is bifid or when its rotation is abnormal, leading to pancreatic tissue surrounding the duodenum.
- Effects: The encircling tissue may produce duodenal obstruction, pancreatitis, or peptic ulceration. In fetal life, severe obstruction may contribute to polyhydramnios.
- Radiological finding: Abdominal imaging may demonstrate the characteristic “double bubble” sign, caused by gas accumulation in the stomach and proximal duodenum.
- Treatment: Management usually involves surgical bypass of the obstructed segment, commonly by duodenojejunostomy.
DEVELOPMENT OF SPLEEN
Development of the Spleen
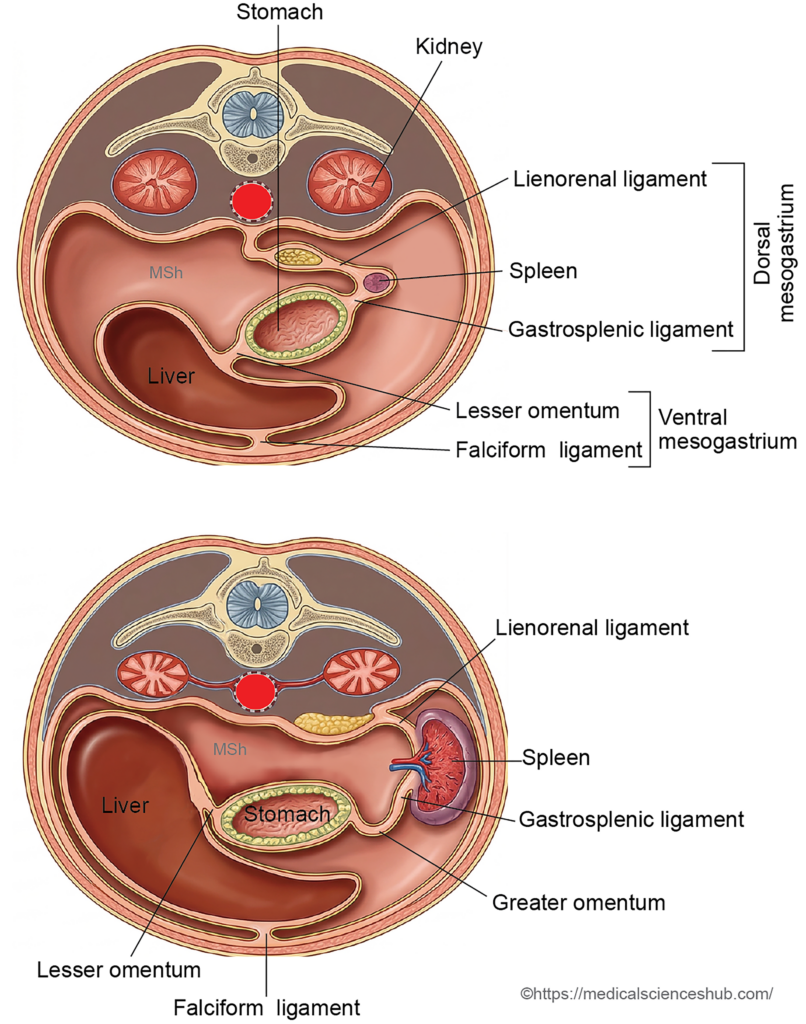
- The spleen develops from mesodermal tissue within the dorsal mesogastrium during early embryonic life.
- As the spleen enlarges, it divides the dorsal mesogastrium into two peritoneal ligaments: the gastrosplenic ligament anteriorly and the lienorenal (splenorenal) ligament posteriorly.
- Mesodermal cells form the structural components of the spleen, including the capsule, septa, connective tissue framework, reticular fibers, lymphocytes, and hematopoietic cells.
Stages of Spleen Development
- Formation of splenic nodules:
Around the 5th week of intrauterine life, clusters of mesenchymal cells appear in the dorsal mesogastrium, forming small nodules called spleniculi or splenic lobules. - Fusion of splenic nodules:
These spleniculi gradually fuse to produce a single splenic mass. - Lobulated pattern:
Early lobulation during development leaves shallow indentations on the adult spleen, visible as splenic notches, typically located along the superior border. - Formation of peritoneal ligaments:
Growth of the spleen separates the dorsal mesogastrium into the gastrosplenic ligament and the lienorenal ligament. The splenic artery runs within the lienorenal ligament. - Final positioning of the spleen:
The spleen reaches its typical location in the left upper abdomen due to rotation of the stomach and fusion of the posterior layer of the dorsal mesogastrium with the posterior abdominal wall. - Role in fetal hematopoiesis:
During fetal life, the spleen functions as an important hematopoietic organ, producing blood cells until near birth. - Genetic regulation:
Development of the spleen is influenced by several genes, including Hox11, Bapx1, and capsulin, which regulate splenic growth and differentiation.
CLINICAL EMBRYOLOGY
Anomalies of Spleen
Abnormal development of the spleen usually results from disturbances in the formation or fusion of splenic nodules (spleniculi) within the dorsal mesogastrium.
- Accessory Spleen (Splenunculus):An accessory spleen develops when one or more spleniculi fail to fuse with the main splenic mass during embryogenesis. These small nodules of functional splenic tissue are relatively common and are usually asymptomatic.
Common locations include:- Hilum of the spleen (most frequent site)
- Gastrosplenic ligament
- Lienorenal (splenorenal) ligament
- Tail of the pancreas
- Along the splenic artery
- Occasionally in the left spermatic cord
- Lobulated Spleen: A lobulated spleen results from incomplete fusion of spleniculi, producing a persistent fetal lobulated appearance of the spleen. This variation is usually identified incidentally and has little clinical significance.
- Splenic Abnormalities in Laterality Disorders: Disorders of left–right body patterning may affect splenic development.
- Right-sided isomerism (bilaterality) is commonly associated with asplenia or a markedly hypoplastic spleen.
- Left-sided isomerism is typically associated with polysplenia, in which multiple small spleens are present.
Important Questions
- Describe the embryological development of the liver.
- Outline the development of the gallbladder.
- Explain the formation of the extrahepatic biliary apparatus.
- Describe the development of the pancreas.
- Enumerate and briefly describe the developmental anomalies of the pancreas.
- Describe the embryological basis, features, and clinical significance of annular pancreas.
- Describe the development of the spleen.

