Competencies
- AN25.3: Describe fetal circulation and changes occurring at birth.
- AN25.5: Describe development basis of congenital anomalies, transportation of great vessels, dextracardia, patient ductus arteriosus, and coarctation of aorta.
- AN25.6: Mention development of aortic arch arteries, SVC, IVC and coronary sinus.
INTRODUCTION
- The development of blood vessels begins during the third week of embryonic development. This process occurs through two main mechanisms: vasculogenesis and angiogenesis.
- Vasculogenesis refers to the de novo formation of blood vessels from mesenchymal cells within the embryo. In this process, mesenchymal cells differentiate into endothelial cells, which then organize to form the initial vascular channels.
- Angiogenesis is the formation of new vessels from pre-existing blood vessels. It occurs through endothelial budding and sprouting, allowing the vascular network to expand into adjacent tissues.
- The earliest vascular structures develop in the extraembryonic mesoderm, particularly in the yolk sac, connecting stalk, and chorion.
- The yolk sac is the first site of blood cell formation and functions as an early hematopoietic organ during embryonic life. This activity continues until approximately 60 days of development.
- As development progresses, the role of blood cell production gradually shifts to other organs, including the liver, spleen, thymus, and eventually the bone marrow, which becomes the primary site of hematopoiesis after birth.
DEVELOPMENT OF ARTERIAL SYSTEM
- By the third week of embryonic development, the primitive circulatory system is established, and several major vascular components are identifiable.
- The heart tube acts as the initial pumping structure of the embryonic cardiovascular system.
- The arterial system includes the following vessels:
- The aortic end of the heart tube, which forms the primary arterial outflow tract.
- The aortic arch arteries, which connect the aortic end of the heart tube to the paired dorsal aortae.
- Two dorsal aortae, located anterior to the notochord, which later contribute to the formation of the descending aorta.
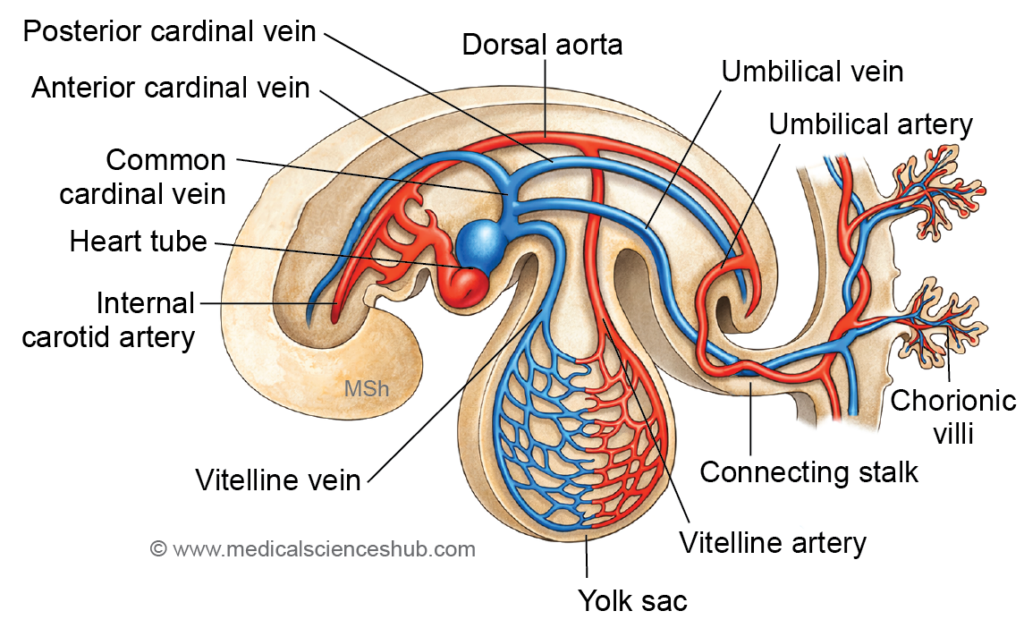
- Vitelline arteries, which supply blood to the yolk sac.
- Umbilical arteries, which transport blood from the embryo to the placenta.
- The venous system is represented by:
- The venous end of the heart tube, which receives returning blood.
- The common cardinal veins, which drain blood from the embryo into the heart through the anterior cardinal veins and posterior cardinal veins.
- The vitelline veins, which return blood from the yolk sac.
- Two umbilical veins, which carry oxygenated blood from the placenta to the embryo.
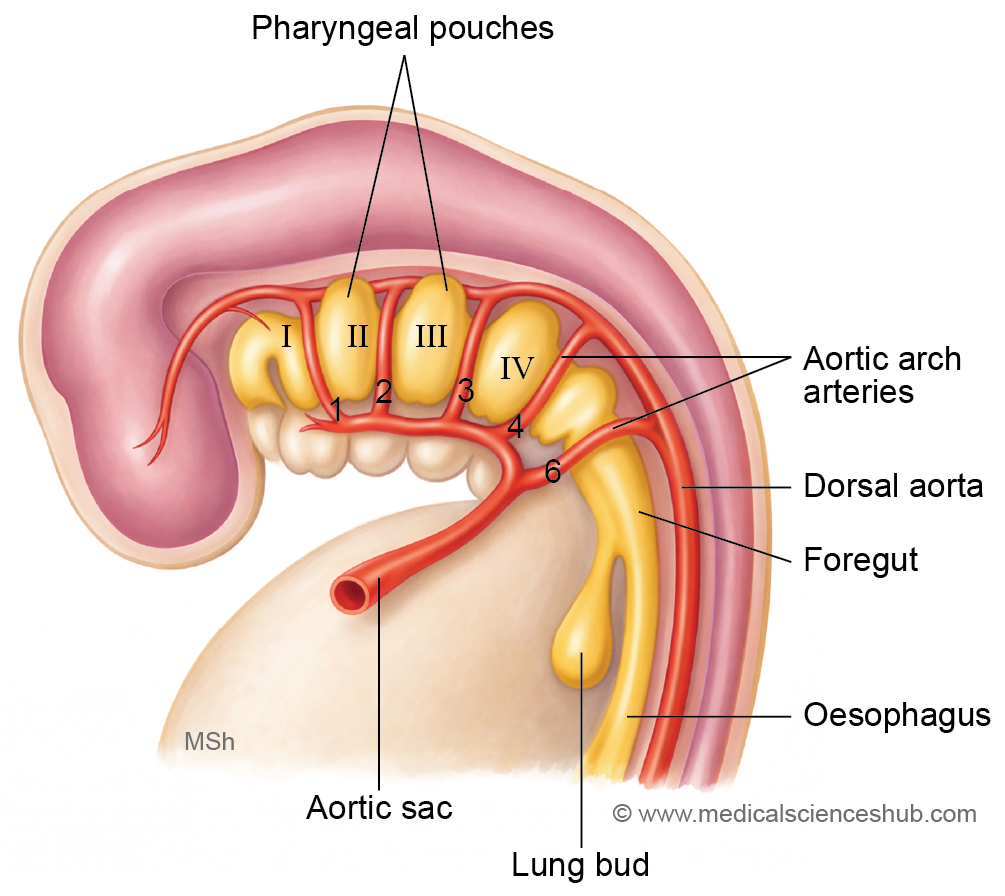
PHARYNGEAL ARCH ARTERIES
- Two primitive aortae arise from the arterial end of the heart tube. These vessels curve dorsally toward the notochord and extend caudally along the length of the embryo.
- Based on their course, the primitive aortae are divided into three regions:
- Ventral aortae, located ventral to the foregut.
- Dorsal aortae, situated dorsal to the foregut and running longitudinally on either side of the embryo.
- The first aortic arch artery, which connects the ventral and dorsal aortae and passes through the first pharyngeal arch.
- The ventral aortae fuse in the midline near the heart region. A dilated portion of this fused segment forms the aortic sac, while the unfused proximal portions contribute to the right and left horns of the aortic sac.
- Each pharyngeal arch receives an artery known as a pharyngeal arch artery. These vessels connect the aortic sac to the dorsal aorta and supply the developing arches. The pharyngeal arches and their arteries appear during the fourth and fifth weeks of development.
- Initially, six pairs of aortic arch arteries develop. However, the fifth pair usually regresses and is often absent in many embryos. Consequently, the arch arteries are conventionally numbered I, II, III, IV, and VI in a cranio-caudal sequence.
- Importantly, all six arch arteries do not exist simultaneously, as some regress while others are forming during embryonic development.
Derivatives of Aortic Arch Arteries
The aortic arch arteries undergo extensive remodeling during embryonic development to form major vessels of the head, neck, and thorax.
- First and Second Arch Arteries
- These arches largely regress during development.
- The first arch artery persists partly as the maxillary artery.
- The second arch artery gives rise to the hyoid artery and stapedial artery.
- Third Arch Artery
- Forms the common carotid artery and the proximal part of the internal carotid artery.
- A vascular sprout from this arch develops into the external carotid artery.
- Fourth Arch Artery
- Shows asymmetrical development on the two sides.
- On the left side, it contributes to a portion of the arch of the aorta.
- On the right side, it forms the proximal segment of the right subclavian artery.
- Fifth Arch Artery
- This arch is usually absent or transient and typically regresses without forming any major adult vessel.
- Sixth Arch Artery (Pulmonary Arch)
- Develops into components of the pulmonary circulation.
- On the left side, it forms the left pulmonary artery and the ductus arteriosus, which later becomes the ligamentum arteriosum after birth.
- On the right side, it contributes to the right pulmonary artery.
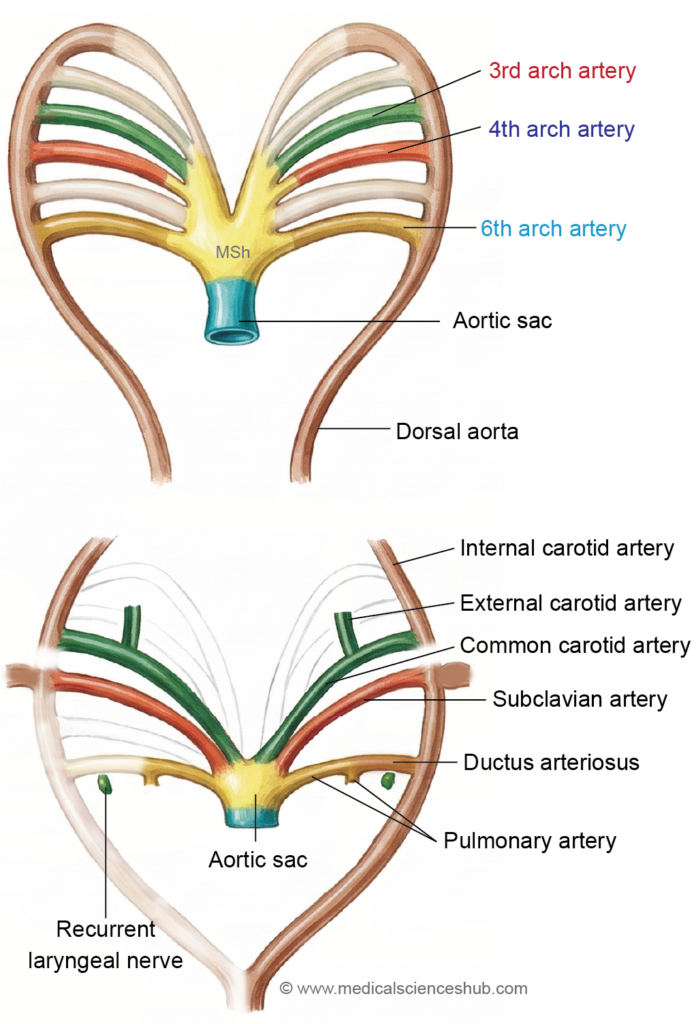
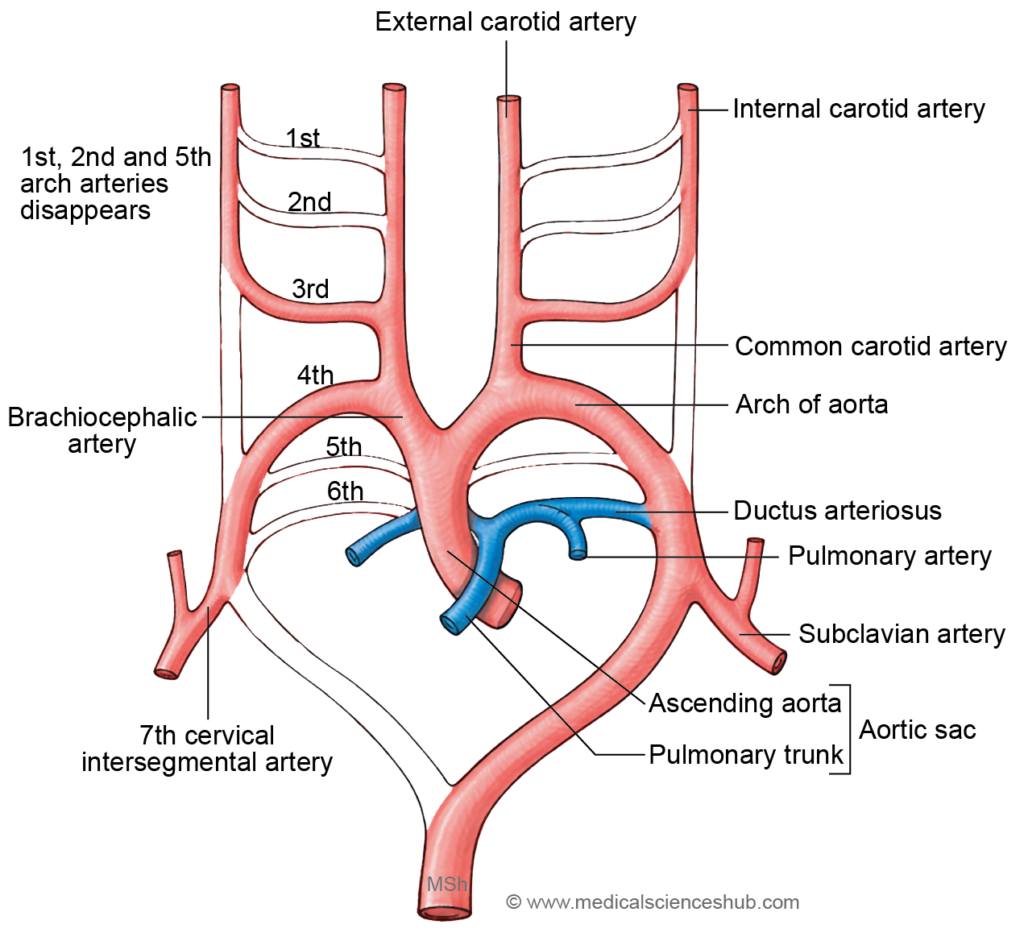
Changes in Arch Arteries and Aortae
- The dorsal aortae remain paired in the region of the pharyngeal arch arteries. Caudal to the sixth arch artery, they fuse to form a single descending aorta, which later develops into the thoracic and abdominal aorta.
- With the formation of the spiral septum in the truncus arteriosus, blood from the ascending aorta is directed into the third and fourth arch arteries, whereas blood from the pulmonary trunk flows into the sixth arch arteries.
- The carotid duct (ductus caroticus) is the segment of the dorsal aorta located between the third and fourth arch arteries. This segment normally regresses completely during development.
Portions That Regress
The following structures normally disappear during vascular remodeling:
- Most parts of the first and second aortic arch arteries
- The carotid duct
- The right dorsal aorta caudal to the fourth arch artery
- The fifth aortic arch (usually absent or transient)
- The distal part of the right sixth arch artery
- The ductus arteriosus (sometimes termed the ductus aorticus) represents the distal portion of the left sixth arch artery, connecting the left pulmonary artery to the left dorsal aorta during fetal life.
Important Developmental Overgrowths
- The dorsal aortae extend cranially beyond the first arch artery and contribute to part of the internal carotid artery.
- A vascular sprout from the third arch artery forms the external carotid artery.
- The seventh intersegmental artery, through its connection with the dorsal aorta near the fourth arch, participates in the formation of the subclavian arteries.
- The proximal portions of the right third and fourth arch arteries contribute to the formation of the brachiocephalic trunk, which arises from the aortic sac.
Table 18.1: Derivatives of pharyngeal arch arteries
| Pharyngeal Arch Artery | Major Adult Derivatives | Key Points |
|---|---|---|
| First (I) | Maxillary artery | Largely regresses; small part persists as maxillary artery supplying deep facial structures. |
| Second (II) | Stapedial, hyoid arteries | Mostly transient; stapedial artery regresses, leaving minor middle ear branches. |
| Third (III) | Common carotid, proximal internal carotid | Forms main carotid system; external carotid arises as a sprout. |
| Fourth (IV) | Left: Aortic arch part; Right: Proximal right subclavian | Asymmetrical development; key contributor to systemic circulation. |
| Fifth (V) | None (absent/regresses) | Often does not form; if present, rudimentary and disappears. |
| Sixth (VI) | Left: Pulmonary artery + ductus arteriosus; Right: Pulmonary artery | Pulmonary arch; ductus arteriosus becomes ligamentum arteriosum after birth. |
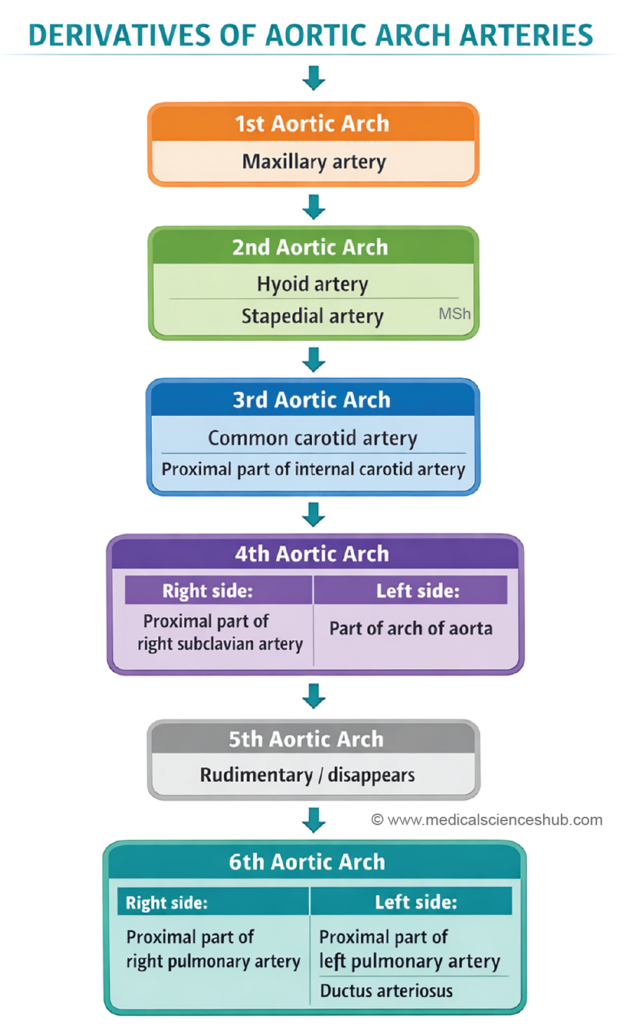
Table 18.2: Development of major arteries
| Adult Artery | Principal Embryonic Origin |
|---|---|
| Arch of the aorta | Develops from three components: the aortic sac (formed by fusion of the ventral aortae), the left horn of the aortic sac, and the left fourth aortic arch artery. |
| Brachiocephalic trunk | Arises from the right horn of the aortic sac, representing the unfused right portion of the ventral aorta. |
| Right subclavian artery | Forms from multiple segments: the right fourth aortic arch artery, a portion of the right dorsal aorta, and the right seventh cervical intersegmental artery, which contributes to the distal part. |
| Left subclavian artery | Derived mainly from the left seventh cervical intersegmental artery. |
| Pulmonary arteries | Originate from the proximal part of the sixth aortic arch artery, also known as the pulmonary arch. |
| Descending aorta | The proximal segment arises from the left dorsal aorta distal to the fourth arch artery, while the distal portion develops from the fused dorsal aortae. |
| Common carotid artery | Develops from the proximal segment of the third aortic arch artery, located before the origin of the external carotid artery. |
| External carotid artery | Arises as a vascular sprout (bud) from the third aortic arch artery during early arterial remodeling. |
| Internal carotid artery | Formed from two embryonic components: the distal part of the third aortic arch artery and the cranial extension of the dorsal aorta beyond the third arch. |
CLINICAL EMBRYOLOGY
Patent Ductus Arteriosus (PDA)
- Embryological Development: During fetal life, the lungs are not involved in oxygen exchange. To bypass pulmonary circulation, several temporary vascular channels are present. One of these is the ductus arteriosus, a short vascular connection that joins the pulmonary trunk with the descending aorta.
- Embryologically, the ductus arteriosus develops from the distal portion of the left sixth aortic arch.
- It allows blood leaving the right ventricle to pass directly into the systemic circulation instead of entering the lungs.
- Normal Changes After Birth: At birth, several physiological changes occur:
- The newborn begins to breathe, causing the lungs to expand.
- Pulmonary vascular resistance decreases.
- Oxygen concentration in the blood rises.
- Prostaglandin levels decrease.
- These changes cause contraction of the ductus arteriosus, leading to functional closure within the first one to two days after birth. Over the following weeks, the vessel becomes fibrotic and forms the ligamentum arteriosum.
- Development of Patent Ductus Arteriosus: When the ductus arteriosus fails to close after birth, the condition is called Patent Ductus Arteriosus (PDA). Because systemic arterial pressure is higher than pulmonary arterial pressure after birth, blood flows from the aorta into the pulmonary artery, producing a left-to-right shunt.
- Pathophysiological Effects: The abnormal shunting of blood increases the volume of blood entering the pulmonary circulation. This produces:
- Pulmonary overcirculation
- Enlargement of the left atrium and left ventricle
- Increased workload on the heart
- Progressive pulmonary hypertension if untreated
- In severe cases, chronic pulmonary hypertension may eventually reverse the direction of blood flow, producing Eisenmenger syndrome.
- Clinical Features: Typical findings include:
- A continuous “machinery-like” heart murmur
- Bounding peripheral pulses
- Wide pulse pressure
- Signs of congestive heart failure in infants
- Management: Treatment depends on age and severity. Medical therapy using prostaglandin inhibitors such as indomethacin or ibuprofen may promote closure in premature infants. If the duct remains open, closure can be achieved through catheter-based devices or surgical ligation.
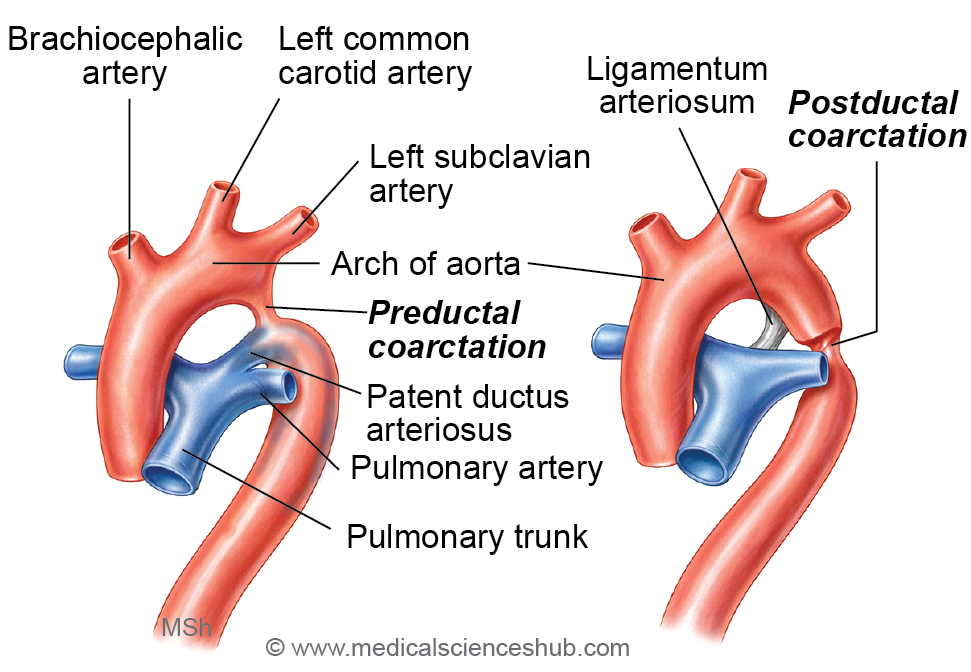
Coarctation of Aorta
- Embryological Development: The aortic arch and descending aorta develop from several embryonic vascular segments, including the fourth aortic arch and portions of the dorsal aorta. The region near the ductus arteriosus is particularly important because it represents a junction between these embryonic vessels.
- Nature of the Defect: Coarctation of the aorta refers to a congenital narrowing of the aortic lumen, most commonly occurring in the region close to the attachment of the ductus arteriosus (later forming the ligamentum arteriosum). The narrowing restricts blood flow to the lower part of the body.
- Types of Coarctation
- Preductal (Infantile) Type
In this form, the narrowing occurs proximal to the ductus arteriosus. Blood supply to the descending aorta depends largely on blood passing through a patent ductus arteriosus. This type often becomes symptomatic early in life. - Postductal (Adult) Type
In the adult type, the narrowing occurs distal to the ductus arteriosus. Over time, collateral circulation develops through enlarged arteries, particularly the intercostal and internal thoracic arteries, to supply blood to the lower body.
- Preductal (Infantile) Type
- Hemodynamic Consequences because of the narrowing:
- Blood pressure becomes elevated in vessels supplying the head and upper limbs
- Blood pressure in the lower limbs is reduced
- The left ventricle must generate greater force to pump blood through the constricted aorta
- Long-standing obstruction may result in left ventricular hypertrophy.
- Clinical Manifestations:
- High blood pressure in the arms
- Weak or delayed femoral pulses
- Lower blood pressure in the legs
- Rib notching on chest radiographs due to enlarged collateral intercostal arteries
- Treatment: Management may involve surgical correction of the narrowed segment or balloon angioplasty with stent placement.
Unusual Origin of Aorta
- Double aortic arch: This anomaly arises from persistence of the right dorsal aorta distal to the seventh cervical intersegmental artery, producing two arches that may encircle the trachea and esophagus.
- Right aortic arch: This condition results from regression of the left dorsal aorta distal to the seventh intersegmental artery, while the corresponding segment of the right dorsal aorta persists.
- Aberrant origin of the right subclavian artery:
- The right subclavian artery may arise directly from the descending aorta due to regression of the right fourth aortic arch artery.
- This anomaly is associated with absence of the brachiocephalic trunk, origin of the right common carotid artery directly from the aortic arch, and loss of the typical recurrent course of the right recurrent laryngeal nerve.
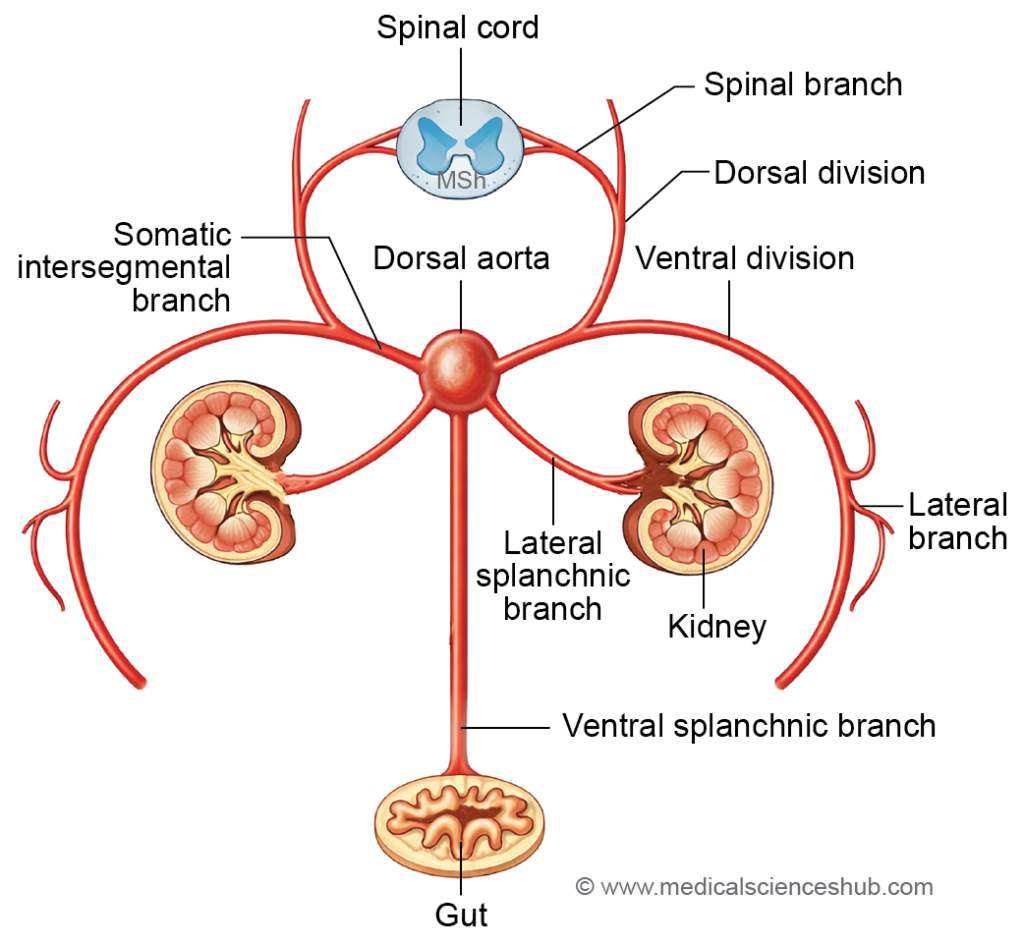
Branches of Dorsal Aorta
The dorsal aorta gives rise to several arterial branches that supply the developing body wall and internal organs. These branches are arranged into three main groups.
1. Dorsolateral branches (Somatic intersegmental arteries)
These arteries supply the body wall and limbs. They develop into:
- Arteries of the limbs
- Intercostal arteries supplying the thoracic wall
- Lumbar arteries supplying the posterior abdominal wall
- Lateral sacral arteries supplying structures in the sacral region
2. Lateral splanchnic branches
These vessels primarily supply developing organs of the urogenital system and adjacent structures. They form the:
- Phrenic arteries
- Suprarenal arteries
- Renal arteries
- Gonadal arteries (testicular or ovarian arteries)
3. Ventral splanchnic branches
These arteries supply the developing gastrointestinal tract and associated organs. They are divided into two groups:
- Vitelline arteries, which give rise to the celiac trunk, superior mesenteric artery, and inferior mesenteric artery.
- Umbilical arteries, which later form the superior vesical arteries, while their distal portions persist as the medial umbilical ligaments after birth.
The caudal remnant of the dorsal aorta below the sacral region persists as the median sacral artery.
Cervical Longitudinal Anastomotic Channels
In the cervical region, the intersegmental arteries are interconnected by longitudinal arterial channels that form during embryonic development. These connections create a continuous vascular network along the neck.
Three principal longitudinal anastomotic channels are formed:
- Precostal anastomosis:
This channel connects adjacent intersegmental arteries anterior to the necks of the ribs. It contributes to the formation of vessels located along the anterior aspect of the cervical region. - Postcostal anastomosis:
This anastomotic channel lies between the developing costal elements (ribs) and the transverse processes of the vertebrae. - Post-transverse anastomosis:
This channel is situated posterior to the transverse processes of the cervical vertebrae.
Remnants and derivatives of these cervical intersegmental arterial anastomoses contribute to the formation of several definitive arteries in the neck, as summarized in the corresponding reference table.
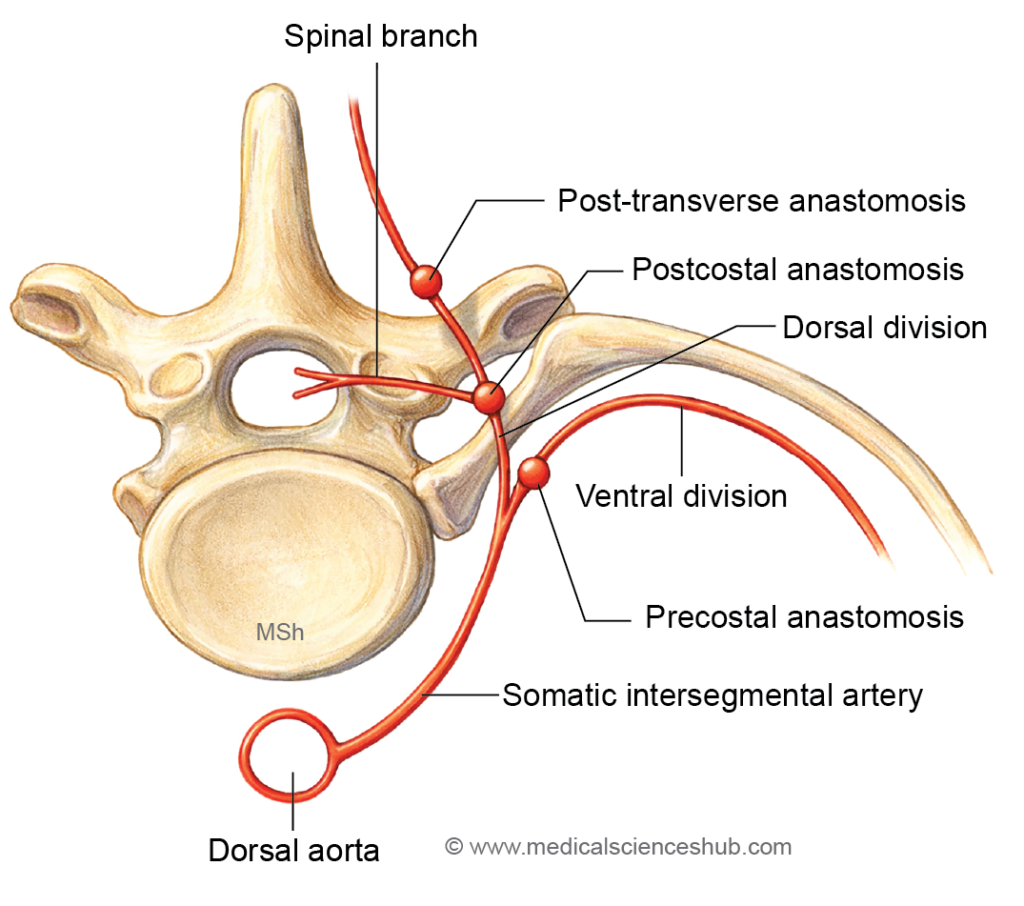
Table 18.3: Derivatives of Cervical Intersegmental Arterial Anastomotic Channels
| Embryonic Anastomotic Channel | Adult Arterial Derivative |
|---|---|
| Precostal anastomotic channel | Contributes to the thyrocervical trunk and gives rise to the ascending cervical artery and superior intercostal arteries. |
| Postcostal anastomotic channel | Forms part of the vertebral artery, which later contributes to the vertebrobasilar circulation supplying the brain. |
| Post-transverse anastomotic channel | Develops into the deep cervical artery, supplying the deep posterior muscles of the neck. |
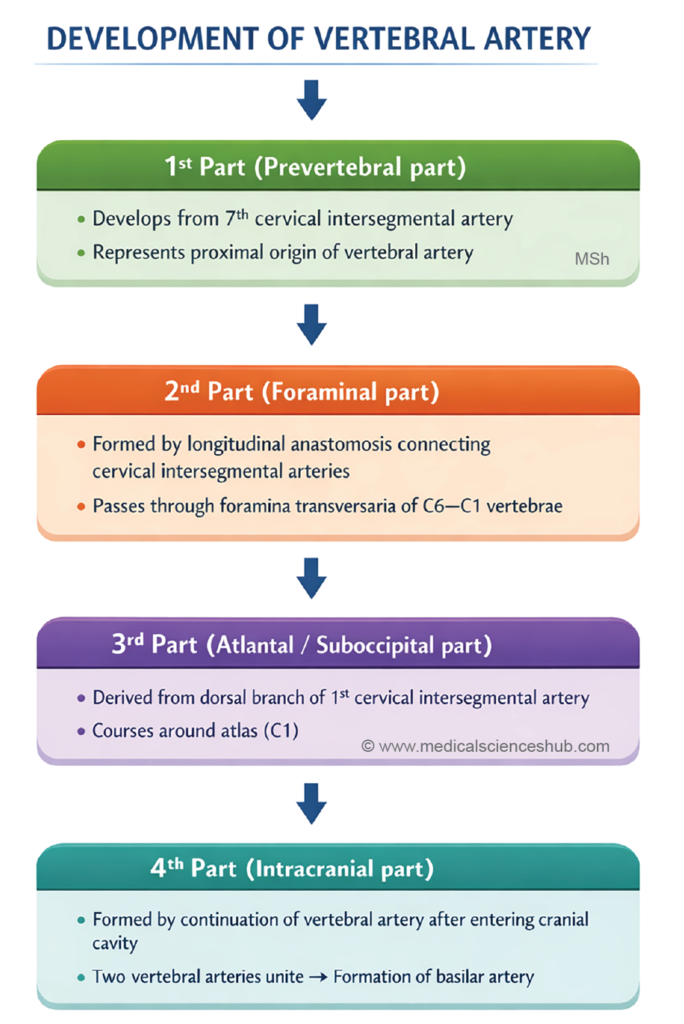
DEVELOPMENT OF VERTEBRAL ARTERY
The vertebral artery develops from longitudinal anastomoses between the cervical intersegmental arteries and is classically described in four anatomical parts.
- First part:
This segment extends from the subclavian artery to the foramen transversarium of the sixth cervical vertebra (C6). It develops from the dorsal division of the seventh cervical intersegmental artery. - Second part:
This portion ascends through the foramina transversaria of the cervical vertebrae from C6 to C1. It forms from the postcostal longitudinal anastomosis connecting the cervical intersegmental arteries. - Third part:
This segment emerges from the foramen transversarium of the atlas (C1) and curves posteriorly to lie on the posterior arch of the atlas before entering the cranial cavity. It develops from the spinal branch of the first cervical intersegmental artery. - Fourth part:
The intracranial segment begins after the artery enters the cranial cavity through the foramen magnum. This part develops from the preneural division of the spinal branch, which contributes to the formation of the basilar arterial system.
UMBILICAL ARTERIES
- The umbilical arteries arise as ventral splanchnic branches of the dorsal aorta during early embryonic development. Each artery plays an essential role in supplying structures associated with the developing placenta and umbilical cord.
- Each umbilical artery provides blood supply to:
- The mesoderm of the connecting stalk
- Structures derived from the allantois
- The developing placenta
- During development, each umbilical artery establishes a connection with a branch of the fifth lumbar artery, which later contributes to the formation of the internal iliac artery. The proximal segment of the umbilical artery between the dorsal aorta and this connection regresses. As a result, the definitive umbilical arteries arise from the internal iliac arteries.
- A single umbilical artery occurs in approximately 1% of births. This anomaly is more frequently observed in twin pregnancies and in infants born to mothers with diabetes. It is often associated with congenital renal abnormalities and other developmental defects.
- Postnatal changes: After birth, the proximal part of each umbilical artery persists as the superior vesical artery, which supplies the urinary bladder. The distal portion becomes fibrotic and forms the medial umbilical ligament.
DEVELOPMENT OF LIMB VESSELS
During embryonic development, each limb is initially supplied by a primary axis artery that runs along the central axis of the limb bud. This artery originates from the intersegmental arteries and forms the principal vascular channel of the developing limb.
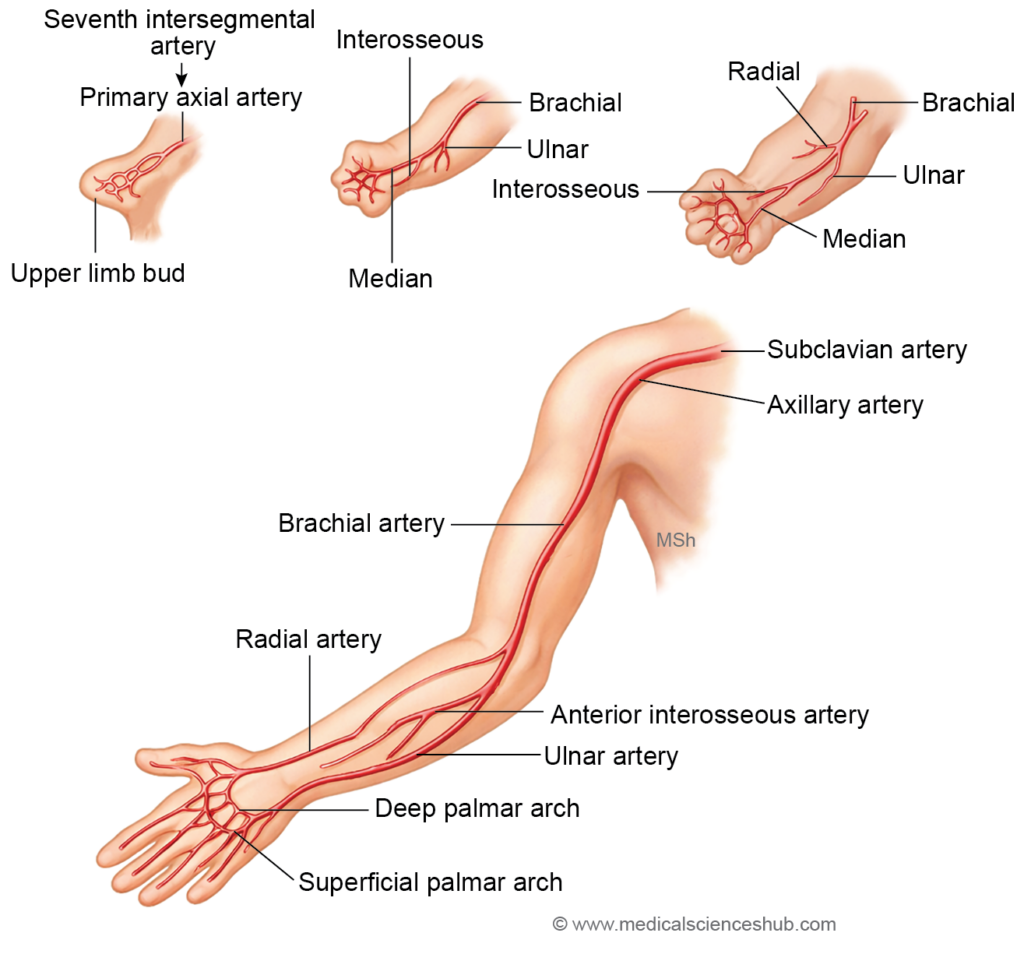
Axis Artery of Upper Limb
The axis artery of the upper limb develops from the seventh cervical intersegmental artery. As development proceeds, this artery gives rise to several major vessels of the upper limb, including:
- Axillary artery
- Brachial artery
- Anterior interosseous artery
- Deep palmar arch
The radial and ulnar arteries appear later as secondary branches that sprout from the axis artery.
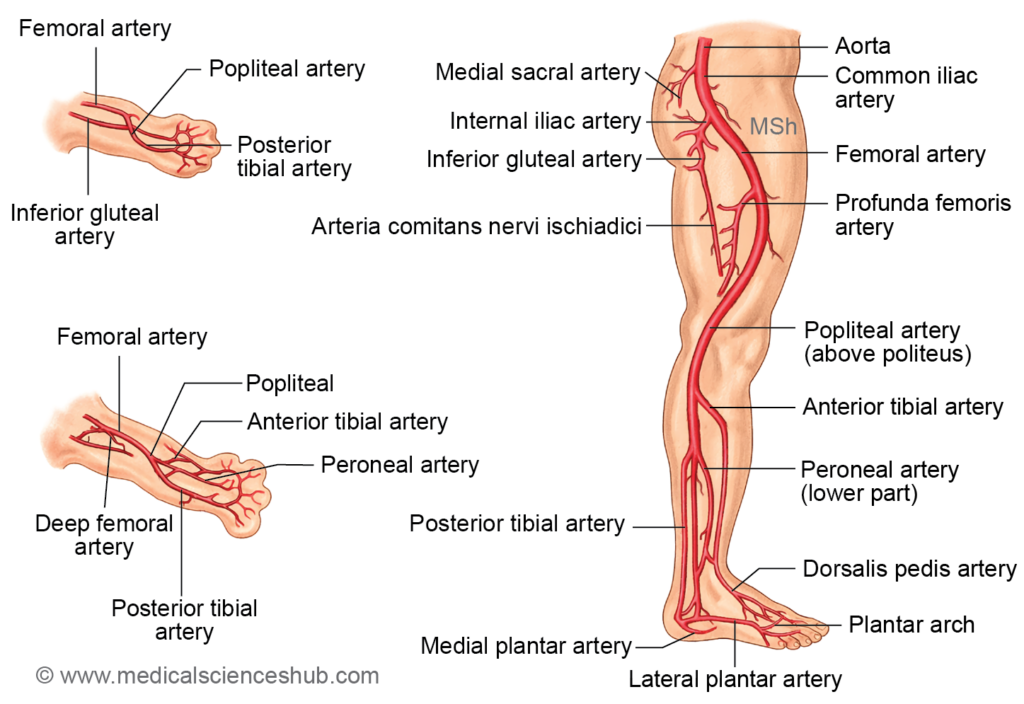
Axis Artery of Lower Limb
The axis artery of the lower limb arises from the fifth lumbar intersegmental artery. It initially forms the main arterial supply to the developing lower limb and later contributes to:
- Arteria comitans nervi ischiadici (the ischiadic artery accompanying the sciatic nerve)
- The proximal part of the popliteal artery above the popliteus muscle
- The distal segment of the peroneal (fibular) artery
- A portion of the plantar arch
The femoral artery develops separately from a capillary plexus on the ventral surface of the thigh. This plexus later establishes connections with the external iliac artery proximally and the popliteal artery distally, forming the definitive arterial pathway of the lower limb.
DEVELOPMENT OF VENOUS SYSTEM
- In the developing embryo, the venous system is organized into two major groups: visceral veins and somatic veins. These vessels collect blood from different embryonic structures and ultimately drain into the sinus venosus of the primitive heart.
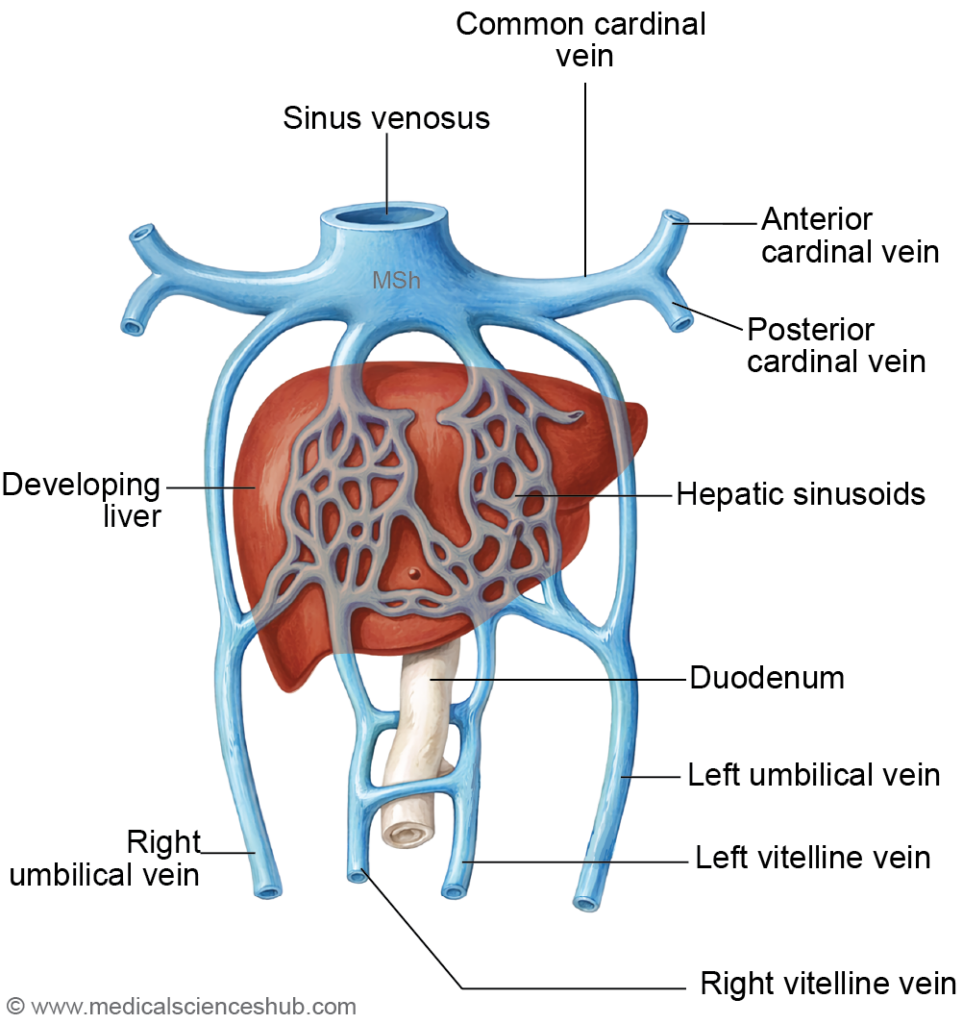
Visceral Veins
- The visceral venous system drains blood from extra-embryonic structures and includes:
- Vitelline (omphalomesenteric) veins, which return blood from the yolk sac.
- Umbilical veins, which carry oxygenated blood from the placenta to the embryo.
H4: Somatic Veins
- The somatic venous system is represented by the cardinal veins, which drain the body of the embryo. The principal components are:
- Anterior cardinal veins (right and left), which drain the cranial region of the embryo and the upper limb buds.
- Posterior cardinal veins (right and left), which drain the caudal part of the embryo and the lower limb buds.
- Common cardinal veins (ducts of Cuvier), formed by the union of the anterior and posterior cardinal veins on each side.
- All these veins ultimately open into the sinus venosus. During later development, these primitive venous channels undergo extensive remodeling and contribute to the formation of the portal venous system, vena cavae, and the azygos venous system.
VITELLINE VEINS
- The embryo initially possesses two vitelline veins (right and left) that return blood from the yolk sac to the heart. With the formation of the head fold, part of the yolk sac becomes incorporated into the primitive gut, and the vitelline veins run on either side of the developing gut tube. They pass through the septum transversum and ultimately drain into the sinus venosus.
- As the liver develops within the septum transversum, the vitelline veins are divided into three segments: infrahepatic, intrahepatic, and suprahepatic parts.
- Infrahepatic part
- The infrahepatic segments lie caudal to the septum transversum on either side of the primitive gut. In the region of the duodenum, the right and left vitelline veins are interconnected by three transverse anastomoses—cranial ventral, middle dorsal, and caudal ventral—forming a characteristic figure-of-eight venous network.
- Important communications occur within this region:
- The left umbilical vein connects with the cranial ventral anastomosis.
- The splenic vein drains into the middle dorsal anastomosis.
- Selective regression of portions of the infrahepatic vitelline veins contributes to the formation of:
- Superior mesenteric vein
- Portal vein trunk
- Right branch of the portal vein
- Left branch of the portal vein.
B. Intrahepatic part
- Within the developing liver, the vitelline veins break up into a capillary plexus that communicates with the hepatic sinusoids. This network differentiates into:
- Afferent venae advehentes, which become branches of the portal vein.
- Efferent venae revehentes, which form tributaries of the hepatic veins.
C. Suprahepatic part
- A subdiaphragmatic anastomosis forms between the right and left vitelline veins and later connects with the cranial ventral intervitelline anastomosis. During further development, the left vitelline vein largely regresses.
- The remaining portions of the suprahepatic vitelline veins give rise to:
- The common hepatic vein, which contributes to the terminal segment of the inferior vena cava (IVC)
- The ductus venosus
- The left hepatic vein
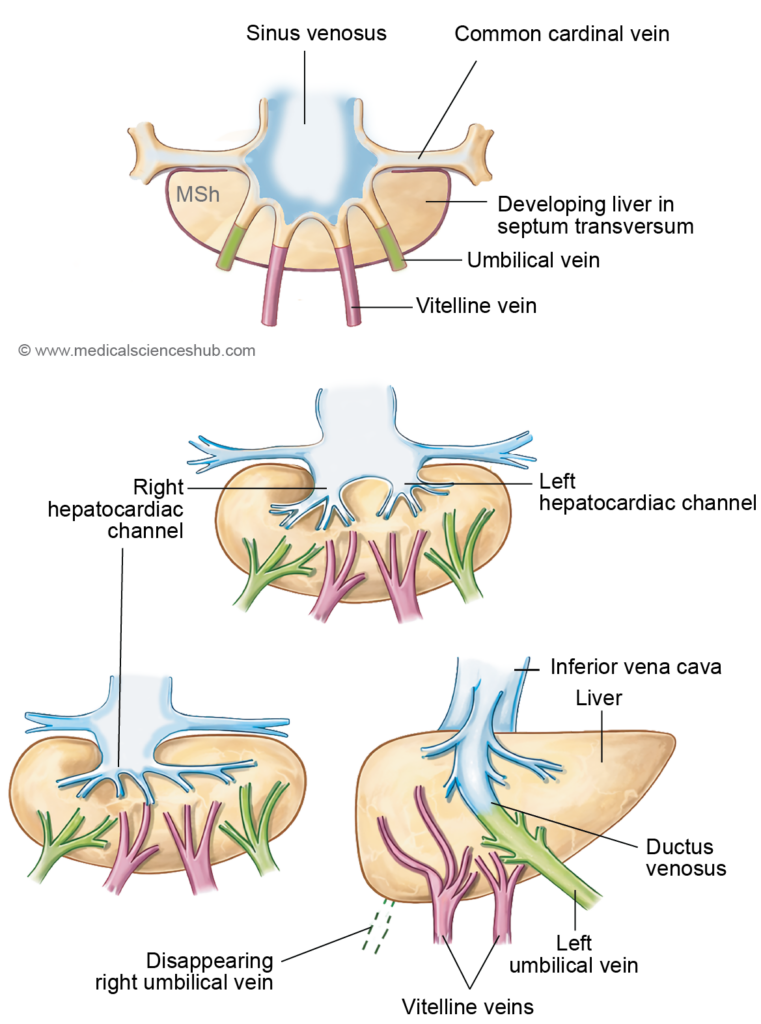
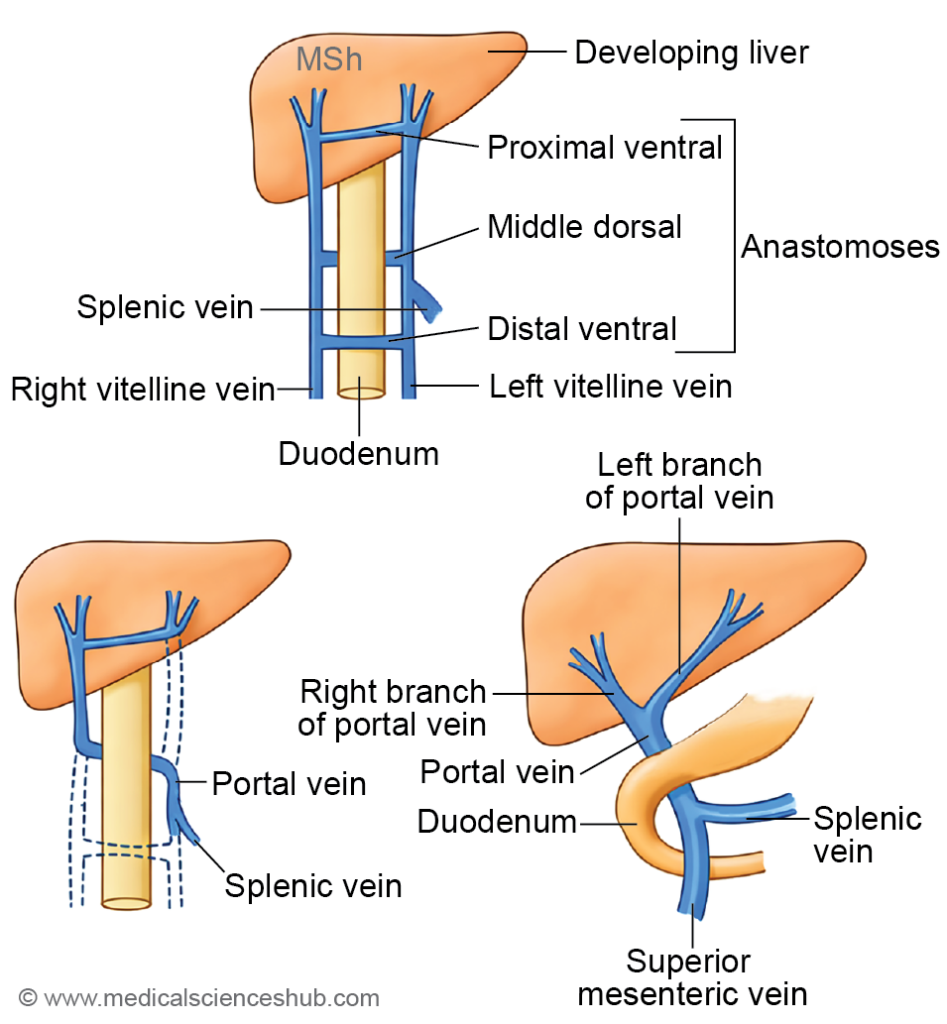
Table 18.4: Development of portal venous system
| Portal Venous Derivative | Embryonic Origin |
|---|---|
| Superior mesenteric vein | Derived from the infrahepatic right vitelline vein distal to the caudal ventral anastomosis, the caudal ventral anastomotic channel, and a segment of the left vitelline vein between the middle dorsal and caudal ventral anastomoses. |
| Trunk of the portal vein | Formed from the middle dorsal anastomosis and the segment of the right vitelline vein between the cranial ventral and middle dorsal anastomoses. |
| Right branch of portal vein | Develops from the infrahepatic portion of the right vitelline vein up to the cranial ventral anastomosis. |
| Left branch of portal vein | Arises from the cranial ventral anastomosis and the infrahepatic left vitelline vein proximal to this anastomosis. |
UMBILICAL VEINS
- The umbilical veins carry oxygenated blood from the placenta to the developing embryo. Initially, two umbilical veins are present, and they pass through the somatopleuric mesoderm and the septum transversum before entering the sinus venosus of the primitive heart.
- During development, the right umbilical vein gradually degenerates, while the left umbilical vein persists and becomes the primary vessel transporting placental blood to the fetus.
- The persistent left umbilical vein establishes a connection with the left vitelline vein, which later contributes to the formation of the left branch of the portal vein. The segment of the left umbilical vein located cranial to the septum transversum subsequently regresses. As a result, blood from the left umbilical vein reaches the heart through two pathways:
- Through the hepatic circulation via the left branch of the portal vein.
- Directly to the inferior vena cava through the ductus venosus, which allows partial bypass of the hepatic sinusoids.
- After birth, the ductus venosus closes and becomes the ligamentum venosum, while the left umbilical vein transforms into the ligamentum teres hepatis.
SOMATIC VEINS
- In the early embryo, two paired somatic venous systems are present:
- Anterior cardinal veins (right and left)
- Posterior cardinal veins (right and left)
- On each side, the anterior and posterior cardinal veins unite to form the common cardinal vein (also called the duct of Cuvier). These common cardinal veins open into the corresponding horns of the sinus venosus. Each anterior cardinal vein consists of two segments: the primary head vein and the cervicothoracic vein.
Primary Head Vein
- The primary head vein lies along the lateral side of the cranial end of the neural tube (developing brain vesicles). It forms two venous plexuses:
- A superficial venous plexus, which develops into the dural venous sinuses
- A deep venous plexus, which forms the cerebral veins
- The dural venous sinuses initially develop as three main stems that drain into the primary head vein:
- Cranial (ventral) dural stem draining the forebrain and midbrain
- Middle dural stem draining the metencephalon
- Caudal dural stem draining the myelencephalon
- Most of the primary head vein later regresses, and a small remaining portion contributes to the formation of the cavernous sinus.
- Communication between the dural stems produces several important sinuses:
- Transverse sinus — channel between the cranial and middle dural stems
- Sigmoid sinus — channel between the middle and caudal dural stems
- Inferior petrosal sinus — connection between the primary head vein and the cavernous sinus
- Superior petrosal sinus — derived from the middle dural stem
- The primary maxillary vein drains into the primary head vein and later develops into the superior ophthalmic vein.
- A sagittal venous plexus forms on the superolateral surface of the forebrain, giving rise to the superior sagittal sinus, straight sinus, and the great cerebral vein.
Cervicothoracic Vein
- The cervicothoracic segment of the anterior cardinal venous system contributes to the formation of several major veins of the neck and thorax:
- Internal jugular vein – develops from the portion of the anterior cardinal vein located cranial to the entry of the subclavian vein.
- Subclavian vein – derived from the seventh intersegmental vein.
- Right brachiocephalic vein – formed from the segment of the right anterior cardinal vein between the right subclavian vein and the oblique intercardinal anastomosis.
- Left brachiocephalic vein – develops from:
- The segment of the left anterior cardinal vein between the left subclavian vein and the oblique intercardinal anastomosis, and
- The oblique intercardinal anastomosis itself.
- Superior vena cava (SVC) – formed from two embryonic components:
- Extrapericardial part: derived from the right anterior cardinal vein caudal to the oblique intercardinal anastomosis.
- Intrapericardial part: derived from the right common cardinal vein.
- Left superior intercostal vein – formed from a small remnant of the left common cardinal vein near the oblique intercardinal channel after most of the left anterior cardinal vein regresses. It is associated with the ligament of the left vena cava.
- Oblique vein of the left atrium (vein of Marshall) – derived from the left common cardinal vein.
- Coronary sinus – develops from the left horn of the sinus venosus.
- In the normal venous system, both the superior vena cava and inferior vena cava primarily originate from right-sided embryonic veins.
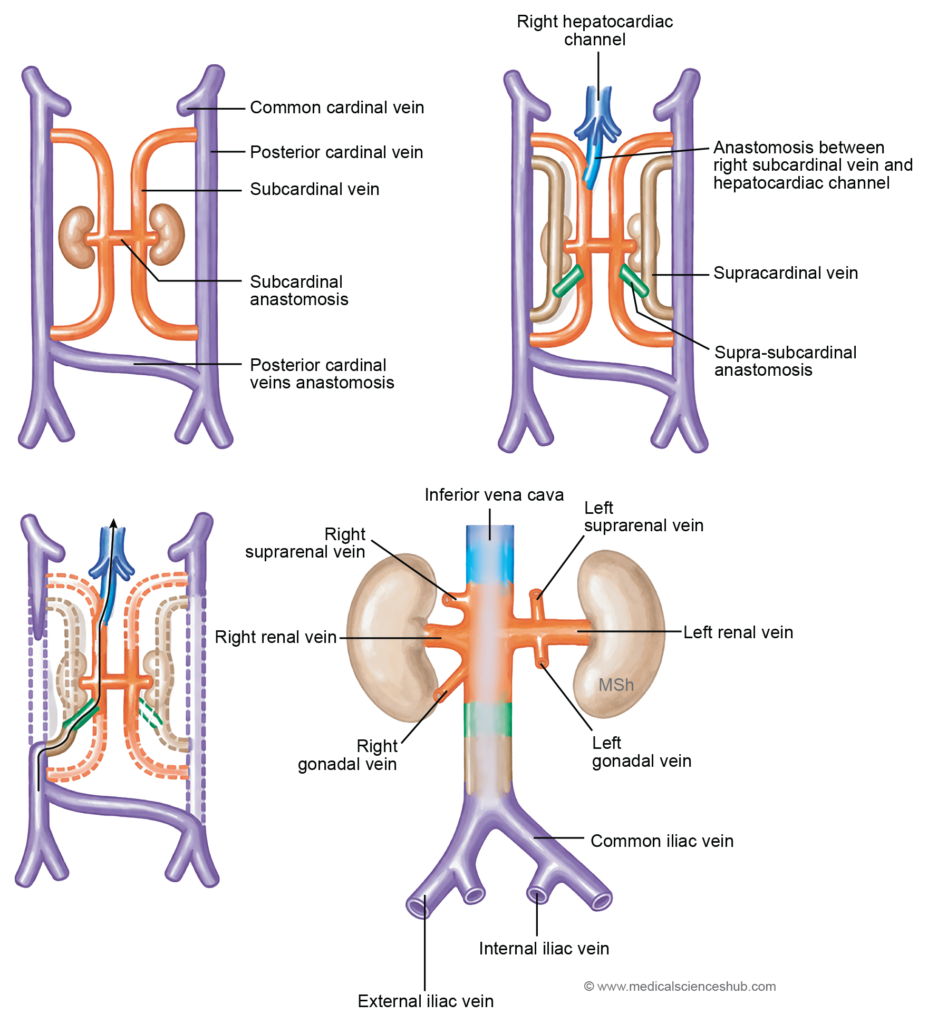
Subcardinal Veins
- The subcardinal veins develop in association with the mesonephric ridge, the region that later contributes to kidney formation. During development, a preaortic anastomosis forms between the right and left subcardinal veins. Each subcardinal vein also establishes cranial and caudal communications with the posterior cardinal vein on the same side.
- A significant connection develops between the right subcardinal vein and the common hepatic vein. This channel is known as the right hepatocardiac channel, which plays an important role in the formation of the inferior vena cava (IVC).
Supracardinal Veins (Thoracolumbar Veins)
- The supracardinal veins develop later than the subcardinal veins and run longitudinally along the dorsolateral aspect of the posterior cardinal veins. Cranially and caudally, they form communications with the corresponding posterior cardinal veins.
- In addition, the supracardinal veins connect with the subcardinal veins through the suprasubcardinal anastomosis, an important channel that contributes to the development of the thoracic and abdominal venous system.
Table 18.5: Subcardinal and supracardinal veins
| Feature | Subcardinal Veins | Supracardinal Veins |
|---|---|---|
| Position | Located ventrolateral to the abdominal aorta during embryonic development. | Situated dorsolateral to the abdominal aorta. |
| Connections (cranial and caudal) | Communicate with the posterior cardinal veins at both upper and lower levels. | Also maintain connections with the posterior cardinal veins. |
| Major Anastomosis | Linked by inter-subcardinal anastomoses, which contribute to venous remodeling. | Participate in the subcardinal–supracardinal anastomosis. |
| Right-side Derivatives | Form the right gonadal vein, right suprarenal vein, and a portion of the inferior vena cava (IVC). | Develop into the postrenal segment of the inferior vena cava. |
| Left-side Derivatives | Give rise to the left gonadal vein and left suprarenal vein. | The left supracardinal vein largely regresses during development. |
Posterior Cardinal Veins
- The paired posterior cardinal veins form an early venous drainage system for the embryonic trunk. Each posterior cardinal vein joins the corresponding anterior cardinal vein to form the common cardinal vein, which drains into the sinus venosus.
- The right and left posterior cardinal veins are interconnected by a transverse iliac anastomosis. This communication contributes to the formation of early pelvic venous channels.
- Each posterior cardinal vein receives several tributaries, including:
- Twelve thoracic intersegmental veins
- Five lumbar intersegmental veins
- Iliac veins, consisting of the internal iliac vein draining the pelvic region and the external iliac vein draining the developing lower limb bud
- During further development of the abdominal venous system, additional paired veins appear, namely the subcardinal veins and supracardinal veins, which gradually replace much of the posterior cardinal system and contribute to the formation of the inferior vena cava and related venous channels.
Azygos Venous Lines
- During embryonic development, longitudinal venous channels appear along the medial side of the sympathetic trunk. Cranially, these channels communicate with the corresponding posterior cardinal veins, while caudally they connect with the subcardinal veins.
- A transverse communication forms between the developing azygos veins, passing dorsal to the aorta. This connection contributes to the establishment of the azygos venous system.
- The developing azygos channels also form multiple communications with the supracardinal veins. As portions of the supracardinal veins regress, these connections persist and give rise to the intersegmental veins, which later differentiate into the intercostal veins and lumbar veins.
Table 18.6: Right and left posterior cardinal veins
| Embryonic Segment | Right Posterior Cardinal Vein – Main Derivatives | Left Posterior Cardinal Vein – Main Derivatives |
|---|---|---|
| Region caudal to the cardinal anastomosis | Contributes to formation of the right common iliac vein and right internal iliac vein. | Forms the left internal iliac vein. |
| Region cranial to the cardinal anastomosis | Most of this segment regresses during development. Remaining parts contribute to the arch of the azygos vein, and the caudal portion helps form the most inferior segment of the inferior vena cava (IVC). | Largely degenerates. The cranial remnant persists as part of the left superior intercostal vein. |
- During embryonic venous remodeling, large portions of the posterior cardinal veins regress, while selected segments persist to form important components of the systemic venous circulation, particularly parts of the iliac veins, the azygos system, and the inferior vena cava.
CLINICAL EMBRYOLOGY
Anomalies of Superior Vena Cava
- Persistent Left Superior Vena Cava
- Embryological basis:Normally, the left anterior cardinal vein regresses during development. If it persists, it forms a left-sided superior vena cava.
- Anatomical features
- The persistent vessel usually drains into the coronary sinus, which then empties into the right atrium.
- The normal right SVC may still be present, producing a double SVC
- Clinical significance
- Often asymptomatic and discovered incidentally.
- May cause dilatation of the coronary sinus.
- Important during cardiac catheterization, pacemaker insertion, and cardiothoracic surgery
- Double Superior Vena Cava
- Embryological basis: Occurs when the left anterior cardinal vein fails to regress while the right SVC develops normally.
- Anatomical features
- Two superior vena cavae are present:
- Right SVC drains into the right atrium normally.
- Left SVC commonly drains into the coronary sinus.
- The left brachiocephalic vein may be absent.
- Two superior vena cavae are present:
- Clinical significance
- Usually asymptomatic.
- Can complicate central venous access and cardiac imaging.
- Important to recognize before cardiac surgery or device implantation.
- Absence of the Superior Vena Cava
- Embryological basis: Results from failure of formation of the right common cardinal vein and right anterior cardinal vein.
- Anatomical features
- Venous return from the upper body is redirected through the azygos venous system.
- Blood eventually reaches the inferior vena cava and right atrium.
DEVELOPMENT OF INFERIOR VENA CAVA
- The inferior vena cava (IVC) develops through a complex sequence of formation, regression, and anastomosis among several embryonic veins, including the posterior cardinal, subcardinal, supracardinal, and vitelline veins.
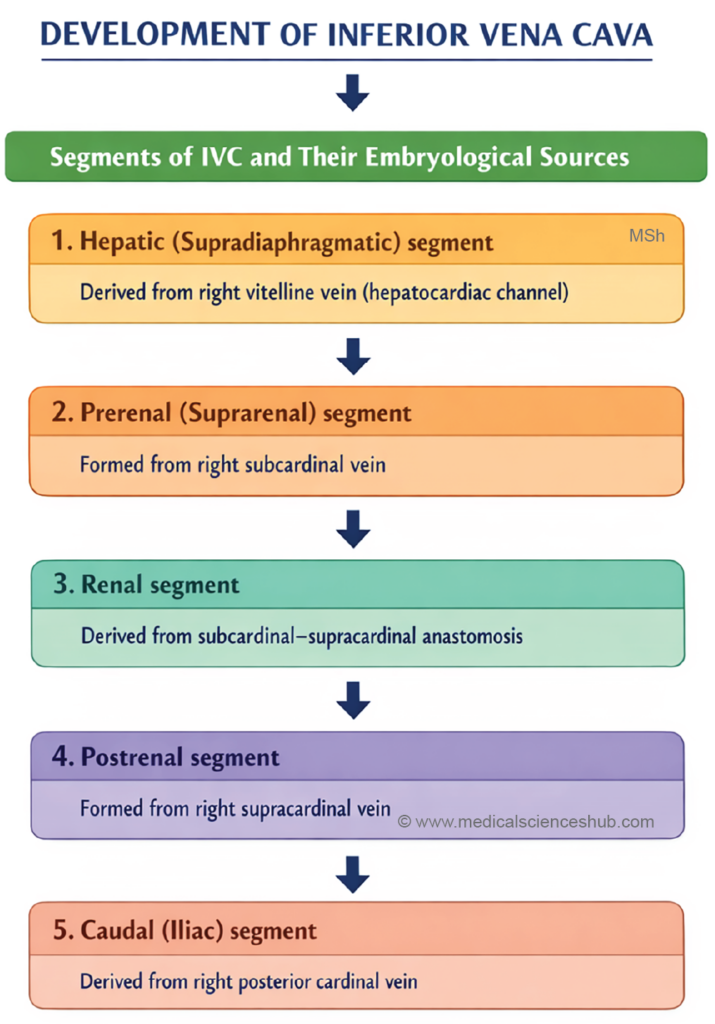
- The different segments of the IVC originate from specific embryonic components:
- Caudal segment – derived from the caudal part of the right posterior cardinal vein, located between the transverse interposterior cardinal anastomosis and the junction where the right supracardinal vein joins the posterior cardinal vein.
- Postrenal segment – develops from the right supracardinal vein.
- Renal segment – formed from the right suprasubcardinal anastomosis, which connects the supracardinal and subcardinal venous systems.
- Prerenal segment – derived from the right subcardinal vein.
- Hepatic segment – develops from an anastomosis between the right subcardinal vein and the right vitelline vein (future common hepatic vein).
- Suprahepatic segment – originates from the right vitelline vein, also known as the right hepatocardiac channel. The corresponding left hepatocardiac channel normally regresses as the left horn of the sinus venosus diminishes during development.
CLINICAL EMBRYOLOGY
Anomalies of the Inferior Vena Cava
- Congenital anomalies of the inferior vena cava arise because its development involves multiple embryonic veins, including the posterior cardinal, subcardinal, and supracardinal veins. Abnormal persistence or regression of these channels can lead to several clinically significant variations.
- Double IVC
- A double inferior vena cava occurs when the left supracardinal vein persists or when proper anastomosis between the paired posterior cardinal veins fails to form. In this condition, an additional left IVC develops from the left posterior cardinal vein, usually below the level of the renal veins.
Clinical significance: It may complicate abdominal surgery, renal transplantation, or placement of IVC filters, and can be misinterpreted as lymphadenopathy on imaging.
- A double inferior vena cava occurs when the left supracardinal vein persists or when proper anastomosis between the paired posterior cardinal veins fails to form. In this condition, an additional left IVC develops from the left posterior cardinal vein, usually below the level of the renal veins.
- Absence of IVC
- Absence or severe hypoplasia of the IVC results from failure of communication between the right subcardinal vein and the right hepatocardiac channel during development.
Clinical significance: Venous blood from the lower body is redirected through collateral pathways such as the azygos and hemiazygos veins, which may become markedly enlarged. This condition may be associated with deep vein thrombosis (DVT) in young individuals.
- Absence or severe hypoplasia of the IVC results from failure of communication between the right subcardinal vein and the right hepatocardiac channel during development.
- Preureteric (Preuretic) IVC
- A preureteric IVC develops when the infrarenal segment of the IVC forms from the subcardinal vein, which lies anterior to the ureter, rather than from the supracardinal vein, which normally lies posterior to it. As a result, the IVC passes anterior to the right ureter.
Clinical significance: The abnormal course of the IVC may compress the ureter and cause hydronephrosis or obstructive uropathy.
- A preureteric IVC develops when the infrarenal segment of the IVC forms from the subcardinal vein, which lies anterior to the ureter, rather than from the supracardinal vein, which normally lies posterior to it. As a result, the IVC passes anterior to the right ureter.
- Azygos Continuation of IVC
- In this anomaly, the hepatic segment of the IVC fails to develop. Venous blood from the lower body is diverted through the azygos vein, which drains into the superior vena cava (SVC). The hepatic veins usually open directly into the right atrium.
Clinical significance: This condition is frequently associated with congenital heart defects and heterotaxy syndromes (e.g., polysplenia syndrome). It is important to recognize during cardiac catheterization and thoracic imaging.
- In this anomaly, the hepatic segment of the IVC fails to develop. Venous blood from the lower body is diverted through the azygos vein, which drains into the superior vena cava (SVC). The hepatic veins usually open directly into the right atrium.
DEVELOPMENT OF AZYGOS VENOUS SYSTEM
- The azygos venous system forms an important collateral pathway for venous return from the thoracic wall and mediastinal structures. It consists of three principal veins: the azygos vein, hemiazygos vein, and accessory hemiazygos vein.
Azygos Vein
The azygos vein develops mainly from:
- The right azygos line vein (derived from the supracardinal system).
- The cranial portion of the right posterior cardinal vein.
The hemiazygos vein arises from:
Hemiazygos Vein
- The lower part of the left azygos line vein.
- The caudal postaortic anastomosis between the right and left azygos line veins.
Accessory Hemiazygos Vein
The accessory hemiazygos vein develops from:
- The upper part of the left azygos line vein.
- The cranial postaortic anastomosis between the right and left azygos line veins.
Together, these veins establish the azygos venous network, which later drains into the superior vena cava and serves as an important alternative pathway for venous return if the inferior vena cava becomes obstructed.
Fetal Circulation
- During intrauterine life, the fetus obtains nutrients and oxygen from the placenta rather than from its own lungs and digestive system. Therefore, the fetus depends on the maternal circulation for the supply of oxygen and nutrients and for the removal of carbon dioxide and metabolic wastes.
Special Structures In Fetal Circulation
- Several specialized structures enable efficient circulation in the fetus and allow blood to bypass organs that are not yet fully functional, particularly the lungs and partially the liver.
- Placenta – The placenta serves as the primary site for exchange of gases, nutrients, and waste products between the maternal and fetal circulations.
- Umbilical vein – The single umbilical vein carries oxygenated and nutrient-rich blood from the placenta to the fetus.
- Umbilical arteries – Two umbilical arteries return deoxygenated blood, carbon dioxide, and metabolic waste from the fetus to the placenta. The oxygen saturation in these arteries is approximately 55–60%.
- Foramen ovale – The foramen ovale is an opening between the right atrium and left atrium that allows most blood entering the right atrium to pass directly to the left atrium, thereby bypassing the nonfunctional fetal lungs.
- Ductus venosus – The ductus venosus carries oxygenated blood from the umbilical vein (via the left branch of the portal vein) to the inferior vena cava, allowing a large portion of blood to bypass the hepatic circulation.
- Ductus arteriosus – The ductus arteriosus connects the left pulmonary artery to the descending aorta, diverting blood away from the lungs and directing it into the systemic circulation.
Peculiarities Of Fetal Circulation
- The fetal circulatory system has several distinctive features that enable efficient oxygen delivery while bypassing organs that are not yet fully functional, particularly the lungs and partially the liver.
- Physiological shunts
Three important vascular shunts allow blood to bypass specific organs: the ductus venosus, foramen ovale, and ductus arteriosus. These channels divert blood away from the liver and lungs, which are not fully functional before birth. - Regulation of oxygenated blood flow
The entry of oxygenated blood from the left umbilical vein into the ductus venosus is regulated by a functional sphincter mechanism, helping control the volume of highly oxygenated blood reaching the fetal circulation. - Mixing of oxygenated and deoxygenated blood
Partial mixing of blood occurs at several sites, including the liver, right and left atria, the terminal part of the inferior vena cava, and the distal portion of the aortic arch. - Trans-septal blood flow
Through the foramen ovale, blood passes from the right atrium to the left atrium, allowing well-oxygenated blood to enter the systemic circulation and supply the brain and upper body. - Preferential blood supply to the upper body
Because relatively well-oxygenated blood is directed toward the ascending aorta, the head and upper limbs receive a richer blood supply during fetal life compared with the lower body.
- Physiological shunts
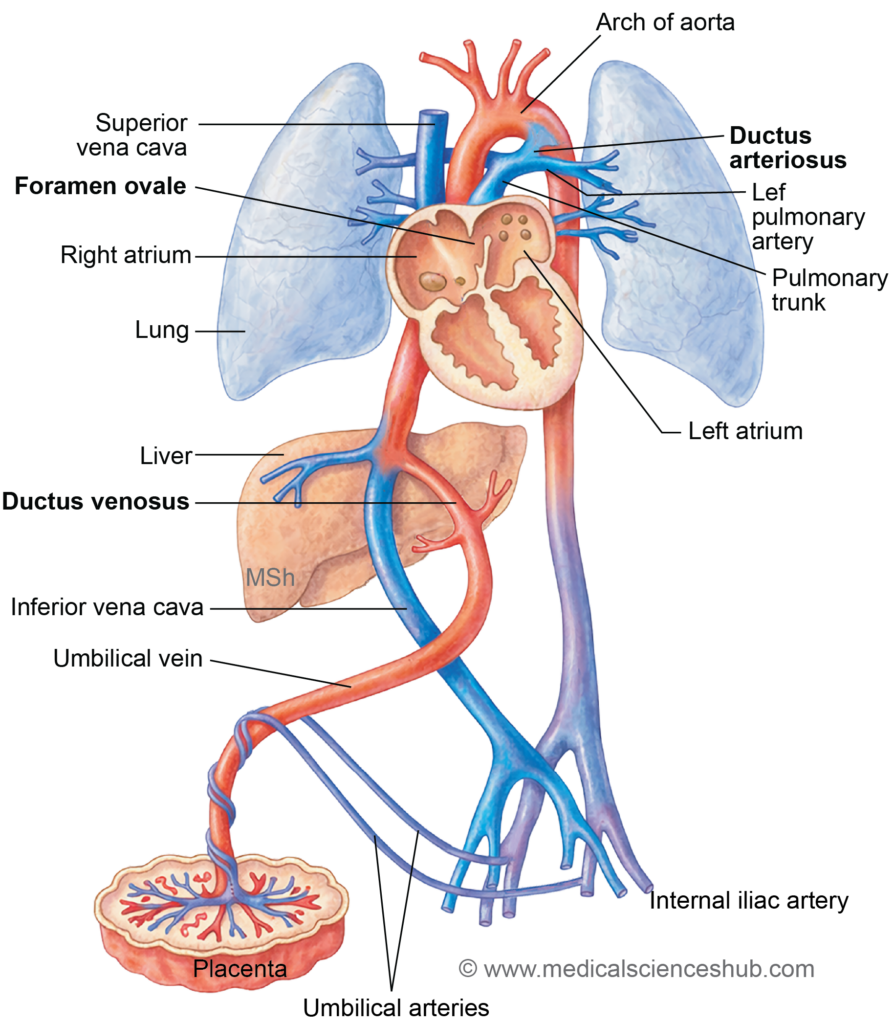

Circulatory Changes At Birth
- At birth, the newborn begins pulmonary respiration, and the lungs replace the placenta as the primary organ for gas exchange. This transition produces several important circulatory changes:
- Closure of umbilical arteries
Contraction of smooth muscle in the umbilical arteries rapidly narrows their lumen, preventing excessive blood loss after the umbilical cord is cut. - Closure of umbilical vein
The umbilical vein constricts slightly later than the arteries. This brief delay allows a small additional volume of placental blood to enter the newborn’s circulation. - Closure of ductus arteriosus
Functional constriction of the ductus arteriosus redirects blood from the right ventricle into the pulmonary circulation, allowing blood to pass through the lungs for oxygenation. - Closure of foramen ovale
Increased blood return from the lungs raises the left atrial pressure, causing the foramen ovale to close functionally as the septum primum presses against the septum secundum. Premature closure during fetal life may lead to right ventricular hypertrophy.
- Closure of umbilical arteries
- Initially, the fetal vascular channels close by vasoconstriction (functional closure). Later, permanent anatomical closure occurs due to proliferation of the tunica intima. Complete obliteration of the umbilical arteries and ductus arteriosus usually occurs within 2–3 months after birth, whereas anatomical closure of the foramen ovale typically occurs by about one year of age.
Table 18.7: Remnants of embryonic vessels
| Embryonic Vessel | Adult Remnant |
|---|---|
| Umbilical arteries | Superior vesical arteries (proximal) and medial umbilical ligaments (distal) |
| Left umbilical vein | Ligamentum teres hepatis |
| Ductus venosus | Ligamentum venosum |
| Ductus arteriosus | Ligamentum arteriosum |
Important Questions
-
Describe the embryological development of the following arteries:
- Arch of the aorta
- Subclavian arteries
- Pulmonary arteries
- Carotid arteries
- Ligamentum arteriosum
- Coarctation of the aorta (embryological basis)
- Explain the embryological basis and types of coarctation of the aorta.
- Describe the embryological development of the vertebral artery.
- Write a short note on the axis artery of the upper limb and lower limb, including its development and adult derivatives.
- Explain the embryological development of the portal vein.
- Describe the formation and developmental origin of the coronary sinus.
- Explain the embryological development of the inferior vena cava.
- Describe fetal circulation and the pathways through which blood flows in the fetus.
- Write a short note on the distinctive features (peculiarities) of fetal circulation.
- Describe the circulatory changes that occur after birth.

